Introduction
-
Minerals are inorganic elements required by the human body for normal growth, development, and maintenance of physiological functions.
-
Unlike carbohydrates, proteins, and fats, minerals do not provide energy, but they are essential for structural, regulatory, and metabolic activities.
-
Minerals are obtained from the diet, as the human body cannot synthesize them.
-
They are present in the body in free ionic form or as components of bones, teeth, enzymes, hormones, and body fluids.
-
Minerals play a vital role in:
-
Formation of bones and teeth
-
Maintenance of fluid and electrolyte balance
-
Nerve conduction and muscle contraction
-
Enzyme activation and metabolism
-
Regulation of acid–base balance
-
-
Based on the amount required by the body, minerals are classified into:
-
Macrominerals (major minerals) – required in amounts >100 mg/day
-
Microminerals (trace elements) – required in amounts <100 mg/day
-
-
Macrominerals constitute 60–80% of the body’s inorganic content, while trace elements, though required in small amounts, are equally essential for life.
-
Minerals act as structural components (calcium, phosphorus), electrolytes (sodium, potassium, chloride), and enzyme cofactors (iron, zinc, copper, manganese).
-
Both deficiency and excess of minerals can lead to serious metabolic and clinical disorders.
General Functions of Minerals
1. Structural Functions
-
Minerals such as calcium, phosphorus, and magnesium are major components of bones and teeth.
-
They provide rigidity, strength, and structural integrity to the skeletal system.
-
Calcium phosphate in the form of hydroxyapatite is responsible for bone hardness.
2. Maintenance of Fluid and Electrolyte Balance
-
Sodium, potassium, and chloride regulate osmotic pressure and acid–base balance.
-
These minerals help maintain intracellular and extracellular fluid distribution.
-
Proper electrolyte balance is essential for normal cellular function.
3. Role in Nerve Transmission and Muscle Contraction
-
Sodium and potassium are essential for generation and propagation of nerve impulses.
-
Calcium and magnesium play a key role in muscle contraction and relaxation.
-
Imbalance can lead to muscle cramps, weakness, or paralysis.
4. Enzyme Activation and Cofactor Functions
-
Many minerals act as enzyme cofactors, enhancing catalytic activity.
-
Examples include:
-
Magnesium for kinases and ATP-dependent enzymes
-
Zinc for dehydrogenases and DNA/RNA polymerases
-
Iron and copper in oxidoreductase enzymes
-
5. Role in Oxygen Transport and Storage
-
Iron is a vital component of hemoglobin and myoglobin.
-
It facilitates oxygen transport in blood and oxygen storage in muscles.
-
Iron deficiency leads to reduced oxygen-carrying capacity.
6. Hormone Synthesis and Action
-
Iodine is essential for synthesis of thyroid hormones (T₃ and T₄).
-
Minerals such as zinc and chromium influence insulin action and glucose metabolism.
-
Some minerals regulate hormone receptor function.
7. Acid–Base Balance
-
Minerals such as phosphates, bicarbonates, and chloride ions help maintain blood pH.
-
They act as buffer systems to prevent drastic changes in hydrogen ion concentration.
8. Blood Clotting (Coagulation)
-
Calcium (Factor IV) is essential for multiple steps in the coagulation cascade.
-
Deficiency may result in delayed clotting and bleeding tendencies.
9. Antioxidant Defense
-
Trace minerals like selenium, zinc, copper, and manganese are components of antioxidant enzymes.
-
Examples include:
-
Glutathione peroxidase (selenium)
-
Superoxide dismutase (zinc, copper, manganese)
-
-
These protect cells from oxidative stress.
10. Growth, Development, and Immunity
-
Minerals such as zinc, iron, and selenium are essential for cell division and immune function.
-
They support wound healing, resistance to infections, and normal growth.
Classification of Minerals
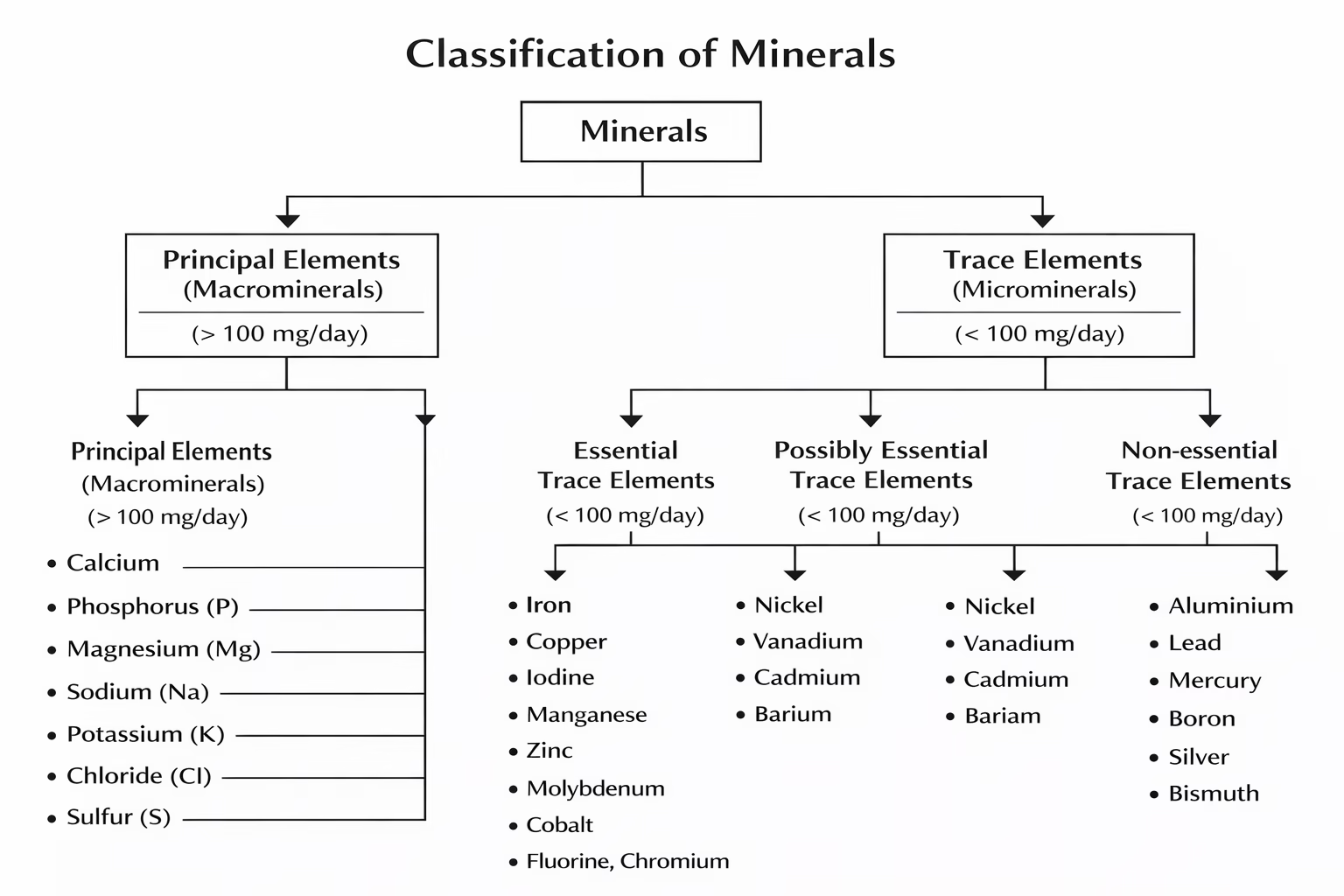
- Macrominerals: Required in amounts greater than 100 mg per day.
- Calcium (Ca)
- Phosphorus (P)
- Magnesium (Mg)
- Sodium (Na)
- Potassium (K)
- Chloride (Cl)
- Microminerals (Trace Minerals): Required in amounts less than 100 mg daily.
- Iron (Fe)
- Zinc (Zn)
- Copper (Cu)
- Manganese (Mn)
- Selenium (Se)
- Iodine (I)
Macrominerals
Calcium
Calcium is the most abundant mineral in the human body, accounting for about 1–2% of total body weight. Nearly 99% of calcium is present in bones and teeth, while the remaining 1% is found in blood, extracellular fluid, and soft tissues, where it plays vital regulatory roles.
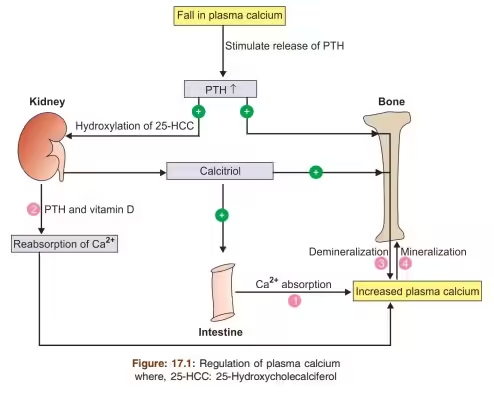
Biochemical Functions of Calcium
A. Structural Functions
-
Major constituent of bones and teeth in the form of hydroxyapatite crystals
(Ca₁₀(PO₄)₆(OH)₂) -
Provides strength, rigidity, and mechanical support to the skeleton
B. Role in Muscle Contraction
-
Essential for excitation–contraction coupling
-
Regulates interaction between actin and myosin
-
Required for contraction of:
-
Skeletal muscle
-
Cardiac muscle
-
Smooth muscle
-
C. Nerve Transmission
-
Facilitates release of neurotransmitters at synaptic junctions
-
Maintains normal nerve excitability
-
Low calcium increases neuronal excitability → tetany
D. Blood Coagulation
-
Calcium acts as Factor IV in the coagulation cascade
-
Required for activation of several clotting factors
E. Enzyme Activation
-
Acts as a cofactor for enzymes such as:
-
ATPase
-
Phospholipase
-
-
Involved in intracellular signaling via calcium–calmodulin complex
F. Hormonal Secretion & Cell Signaling
-
Essential for secretion of hormones (e.g., insulin)
-
Acts as a second messenger in many signal transduction pathways
Dietary Requirements of Calcium
| Age / Condition | Requirement (mg/day) |
|---|---|
| Adults | 1000 mg |
| Adolescents | 1200–1300 mg |
| Pregnant women | 1200 mg |
| Lactating mothers | 1200–1500 mg |
| Elderly | 1200 mg |
Dietary Sources of Calcium
A. Animal Sources
-
Milk and milk products (best source)
-
Milk
-
Curd
-
Cheese
-
Butter milk
-
-
Fish with bones (sardines)
B. Plant Sources
-
Green leafy vegetables (spinach, broccoli)
-
Ragi (finger millet)
-
Sesame seeds
-
Soybeans and tofu
-
Nuts (almonds)
Absorption of Calcium
-
Occurs mainly in the duodenum and jejunum
-
Two mechanisms:
-
Active transport (vitamin D–dependent)
-
Passive diffusion
-
-
About 20–40% of dietary calcium is absorbed
-
Absorption is highest during growth, pregnancy, and lactation
Factors Promoting Calcium Absorption
-
Vitamin D (Calcitriol) – most important factor
-
Adequate gastric acidity
-
Lactose (especially in infants)
-
Optimal Ca : P ratio (1:1 to 2:1)
-
Growth hormone, estrogen
-
Low dietary calcium (adaptive increase in absorption)
Factors Inhibiting Calcium Absorption
-
Phytates (cereals, bran)
-
Oxalates (spinach, chocolate)
-
Excess dietary phosphate
-
Fat malabsorption (formation of calcium soaps)
-
Vitamin D deficiency
-
Alkaline pH of intestine
-
Excess fiber intake
Disease States Related to Calcium
A. Hypocalcemia
(Serum Ca < 8.5 mg/dL)
Causes:
-
Vitamin D deficiency
-
Hypoparathyroidism
-
Chronic kidney disease
-
Malabsorption syndromes
Clinical Features:
-
Tetany
-
Muscle cramps
-
Carpopedal spasm
-
Convulsions
-
Positive Chvostek’s and Trousseau’s signs
B. Rickets
-
Occurs in children
-
Due to vitamin D deficiency
-
Defective mineralization of growing bones
Features:
-
Bow legs
-
Delayed dentition
-
Rachitic rosary
-
Soft skull bones (craniotabes)
C. Osteomalacia
-
Occurs in adults
-
Defective mineralization of bone matrix
Features:
-
Bone pain
-
Fragility fractures
-
Muscle weakness
D. Osteoporosis
-
Common in postmenopausal women and elderly
-
Reduced bone mass with normal mineralization
Risk Factors:
-
Estrogen deficiency
-
Low calcium intake
-
Sedentary lifestyle
E. Hypercalcemia
(Serum Ca > 10.5 mg/dL)
Causes:
-
Hyperparathyroidism
-
Malignancy
-
Excess vitamin D intake
Clinical Features:
-
Kidney stones
-
Polyuria and polydipsia
-
Constipation
-
Neuropsychiatric symptoms
Phosphorus
Phosphorus is the second most abundant mineral in the human body after calcium. About 85% of phosphorus is present in bones and teeth, while the remaining amount is distributed in soft tissues and body fluids. It plays a central role in energy metabolism, nucleic acid synthesis, and acid–base balance.
Biochemical Functions of Phosphorus
A. Structural Functions
-
Present in bones and teeth as calcium phosphate and hydroxyapatite
-
Provides rigidity and strength to skeletal system
B. Role in Energy Metabolism
-
Essential component of ATP, ADP, and AMP
-
Involved in energy storage and transfer
-
Plays a key role in oxidative phosphorylation
C. Component of Nucleic Acids
-
Forms the phosphate backbone of DNA and RNA
-
Essential for cell division and protein synthesis
D. Cell Membrane Structure
-
Constituent of phospholipids (phosphatidylcholine, phosphatidylserine)
-
Maintains membrane integrity and permeability
E. Acid–Base Balance
-
Acts as an important buffer system (phosphate buffer)
-
Helps maintain physiological pH of blood and tissues
F. Enzyme Regulation
-
Required for phosphorylation and dephosphorylation reactions
-
Regulates activity of enzymes and signaling proteins
G. Oxygen Delivery
-
Part of 2,3-bisphosphoglycerate (2,3-BPG) in RBCs
-
Facilitates oxygen release from hemoglobin
Dietary Requirements of Phosphorus
| Age / Condition | Requirement (mg/day) |
|---|---|
| Adults | 700 mg |
| Adolescents | 1250 mg |
| Pregnant women | 700 mg |
| Lactating mothers | 700 mg |
| Elderly | 700 mg |
Dietary Sources of Phosphorus
A. Animal Sources
-
Milk and milk products
-
Meat
-
Fish
-
Poultry
-
Eggs
B. Plant Sources
-
Cereals and whole grains
-
Pulses and legumes
-
Nuts and seeds
-
Soybeans
Note: Phosphorus from plant sources may be less bioavailable due to phytate binding.
Absorption of Phosphorus
-
Absorbed mainly in the duodenum and jejunum
-
Absorption occurs by:
-
Active transport (vitamin D-dependent)
-
Passive diffusion
-
-
About 60–70% of dietary phosphorus is absorbed
-
Absorption is generally efficient compared to calcium
Factors Promoting Phosphorus Absorption
-
Vitamin D (Calcitriol)
-
Adequate dietary intake
-
Growth and increased metabolic demand
-
Normal intestinal function
-
Proper calcium–phosphorus balance
Factors Inhibiting Phosphorus Absorption
-
Excess dietary calcium (forms insoluble Ca-phosphate)
-
Phytates in cereals and legumes
-
Aluminium-containing antacids
-
Vitamin D deficiency
-
Chronic diarrhea or malabsorption
Disease States Related to Phosphorus
A. Hypophosphatemia
(Serum phosphorus < 2.5 mg/dL)
Causes:
-
Vitamin D deficiency
-
Malnutrition
-
Chronic alcoholism
-
Diabetic ketoacidosis (recovery phase)
-
Hyperparathyroidism
Clinical Features:
-
Muscle weakness
-
Bone pain
-
Osteomalacia
-
Rickets in children
-
Hemolysis and impaired oxygen delivery
B. Rickets
-
Occurs in children
-
Due to deficiency of vitamin D, calcium, or phosphorus
-
Leads to defective mineralization of growing bones
Features:
-
Bowing of legs
-
Delayed dentition
-
Rachitic rosary
C. Osteomalacia
-
Occurs in adults
-
Softening of bones due to defective mineralization
Features:
-
Bone pain
-
Fragility fractures
-
Muscle weakness
D. Hyperphosphatemia
(Serum phosphorus > 4.5 mg/dL)
Causes:
-
Chronic kidney disease
-
Hypoparathyroidism
-
Excess intake of phosphate laxatives
-
Tumor lysis syndrome
Clinical Features:
-
Hypocalcemia
-
Soft tissue calcification
-
Renal damage
-
Cardiovascular complications
E. Chronic Kidney Disease–Mineral Bone Disorder (CKD-MBD)
-
Reduced phosphate excretion
-
Secondary hyperparathyroidism
-
Bone demineralization and fractures
Magnesium
Magnesium is an essential macromineral and the fourth most abundant cation in the human body. About 60–70% of magnesium is present in bones, 20–30% in muscles and soft tissues, and <1% in extracellular fluid. It plays a vital role in enzyme activation, energy metabolism, neuromuscular function, and ion transport.
Biochemical Functions of Magnesium
A. Cofactor for Enzymes
-
Acts as a cofactor for more than 300 enzymes
-
Required for enzymes involved in:
-
Glycolysis
-
TCA cycle
-
Oxidative phosphorylation
-
-
Essential for ATP-dependent reactions (Mg-ATP complex)
B. Energy Metabolism
-
Magnesium stabilizes ATP
-
Required for ATP synthesis, storage, and utilization
-
Plays a key role in cellular energy transfer
C. Nucleic Acid and Protein Synthesis
-
Required for DNA and RNA synthesis
-
Essential for ribosomal function and protein synthesis
D. Neuromuscular Function
-
Regulates nerve impulse transmission
-
Controls neuromuscular excitability
-
Acts as a physiological calcium antagonist
E. Muscle Contraction and Relaxation
-
Involved in muscle relaxation
-
Prevents excessive calcium-mediated contraction
-
Deficiency leads to muscle cramps and tremors
F. Bone Mineralization
-
Present in bone matrix
-
Influences bone strength and structure
-
Regulates calcium metabolism
G. Electrolyte Balance
-
Maintains intracellular levels of:
-
Potassium
-
Calcium
-
-
Essential for normal cardiac rhythm
Dietary Requirements of Magnesium
| Age / Condition | Requirement (mg/day) |
|---|---|
| Adult males | 400–420 mg |
| Adult females | 310–320 mg |
| Pregnant women | 350–360 mg |
| Lactating mothers | 310–360 mg |
| Elderly | ~400 mg |
Dietary Sources of Magnesium
A. Plant Sources (Richest)
-
Green leafy vegetables (chlorophyll contains Mg)
-
Whole grains
-
Pulses and legumes
-
Nuts and seeds (almonds, cashews)
-
Bananas
B. Animal Sources
-
Milk and milk products
-
Meat
-
Fish
Absorption of Magnesium
-
Occurs mainly in the small intestine (jejunum and ileum)
-
Absorbed by:
-
Passive diffusion
-
Active transport
-
-
About 30–50% of dietary magnesium is absorbed
-
Kidneys play a major role in magnesium homeostasis
Factors Promoting Magnesium Absorption
-
Adequate dietary intake
-
Vitamin D (to some extent)
-
Normal intestinal function
-
Increased physiological demand
-
Low body magnesium levels
Factors Inhibiting Magnesium Absorption
-
Phytates and oxalates
-
Chronic diarrhea
-
Alcoholism
-
High calcium or phosphate intake
-
Malabsorption syndromes
-
Certain drugs (diuretics, proton pump inhibitors)
Disease States Related to Magnesium
A. Hypomagnesemia
(Serum Mg < 1.5 mg/dL)
Causes:
-
Malnutrition
-
Chronic alcoholism
-
Prolonged diarrhea
-
Diuretics
-
Malabsorption
Clinical Features:
-
Muscle cramps
-
Tremors
-
Tetany
-
Cardiac arrhythmias
-
Seizures
-
Hypocalcemia and hypokalemia
B. Neuromuscular Hyperexcitability
-
Increased nerve firing
-
Muscle spasms
-
Often associated with hypocalcemia
C. Cardiac Arrhythmias
-
Magnesium deficiency predisposes to:
-
Ventricular arrhythmias
-
Torsades de pointes
-
D. Hypermagnesemia
(Serum Mg > 2.5 mg/dL)
Causes:
-
Renal failure
-
Excess intake of magnesium-containing antacids or laxatives
Clinical Features:
-
Muscle weakness
-
Hypotension
-
Bradycardia
-
Respiratory depression
-
Cardiac arrest (severe cases)
Clinical Uses of Magnesium
-
Magnesium sulfate in:
-
Eclampsia
-
Torsades de pointes
-
Severe asthma
-
-
Treatment of hypomagnesemia
Sodium
Sodium is the major extracellular cation and an essential macromineral required for the maintenance of fluid balance, nerve conduction, muscle contraction, and acid–base equilibrium. It plays a central role in osmotic pressure regulation and cellular homeostasis.
Biochemical Functions of Sodium
A. Maintenance of Osmotic Pressure
-
Sodium is the primary determinant of extracellular fluid (ECF) volume
-
Regulates water distribution between intracellular and extracellular compartments
B. Nerve Impulse Transmission
-
Essential for generation and propagation of action potentials
-
Sodium influx causes depolarization of nerve and muscle membranes
C. Muscle Contraction
-
Required for normal muscle excitability
-
Works in coordination with potassium and calcium
D. Acid–Base Balance
-
Sodium salts (NaHCO₃, Na₂HPO₄) act as buffer systems
-
Helps maintain physiological pH
E. Active Transport Mechanisms
-
Sodium gradient drives:
-
Na⁺/K⁺-ATPase pump
-
Secondary active transport of glucose and amino acids
-
F. Regulation of Blood Pressure
-
Influences plasma volume and arterial pressure
-
Excess sodium intake is linked to hypertension
Dietary Requirements of Sodium
| Age / Condition | Requirement |
|---|---|
| Adults | 1500 mg/day |
| Upper safe limit | < 2300 mg/day |
| Pregnant women | 1500 mg/day |
| Lactating mothers | 1500 mg/day |
| Athletes / heavy sweating | Increased requirement |
1 g sodium = 2.5 g common salt (NaCl)
Dietary Sources of Sodium
A. Natural Sources
-
Milk
-
Meat
-
Eggs
-
Vegetables
B. Added / Processed Sources
-
Common salt (NaCl)
-
Pickles
-
Processed foods
-
Canned foods
-
Snacks and fast food
Absorption of Sodium
-
Sodium is rapidly and efficiently absorbed
-
Absorption occurs mainly in the small intestine
-
Mechanisms include:
-
Passive diffusion
-
Active transport
-
Co-transport with glucose and amino acids
-
-
Absorption efficiency is >95%
Factors Promoting Sodium Absorption
-
Presence of glucose and amino acids
-
Normal intestinal mucosa
-
Aldosterone (renal conservation, not intestinal absorption)
-
Increased dietary intake
Factors Inhibiting Sodium Absorption / Causing Loss
-
Chronic diarrhea and vomiting
-
Excessive sweating
-
Diuretics
-
Adrenal insufficiency
-
Intestinal malabsorption disorders
Disease States Related to Sodium
A. Hyponatremia
(Serum Na⁺ < 135 mEq/L)
Causes:
-
Excess water intake
-
Prolonged vomiting or diarrhea
-
Diuretic therapy
-
SIADH (Syndrome of Inappropriate ADH Secretion)
Clinical Features:
-
Nausea and vomiting
-
Headache
-
Confusion
-
Muscle cramps
-
Seizures (severe cases)
B. Hypernatremia
(Serum Na⁺ > 145 mEq/L)
Causes:
-
Water deprivation
-
Excess sodium intake
-
Diabetes insipidus
-
Severe dehydration
Clinical Features:
-
Thirst
-
Dry mucous membranes
-
Irritability
-
Restlessness
-
Coma (severe cases)
C. Hypertension
-
Chronic high sodium intake causes:
-
Increased plasma volume
-
Increased peripheral resistance
-
-
Major risk factor for:
-
Cardiovascular disease
-
Stroke
-
Kidney disease
-
Hormonal Regulation of Sodium Balance
-
Aldosterone: Increases sodium reabsorption in kidneys
-
ADH (Vasopressin): Regulates water balance
-
ANP (Atrial Natriuretic Peptide): Promotes sodium excretion
Potassium
Potassium is the major intracellular cation of the body and an essential macromineral. About 98% of total body potassium is present inside cells, especially in muscle tissues. Potassium is crucial for maintenance of cell membrane potential, nerve impulse transmission, muscle contraction, and cardiac function.
Biochemical Functions of Potassium
A. Maintenance of Resting Membrane Potential
-
Potassium concentration gradient across the cell membrane determines the resting membrane potential
-
Essential for excitability of nerves and muscles
B. Nerve Impulse Transmission
-
Efflux of potassium is responsible for repolarization of nerve membranes
-
Works in coordination with sodium during action potentials
C. Muscle Contraction
-
Required for normal contraction of:
-
Skeletal muscle
-
Cardiac muscle
-
Smooth muscle
-
-
Maintains normal heart rhythm
D. Regulation of Acid–Base Balance
-
Potassium participates in H⁺–K⁺ exchange mechanisms
-
Helps maintain intracellular pH
E. Enzyme Activation
-
Activates several enzymes involved in:
-
Carbohydrate metabolism
-
Protein synthesis
-
F. Cellular Metabolism
-
Facilitates glycogen synthesis
-
Influences insulin secretion and action
Dietary Requirements of Potassium
| Age / Condition | Requirement |
|---|---|
| Adults | 3500–4700 mg/day |
| Pregnant women | 4700 mg/day |
| Lactating mothers | 5100 mg/day |
| Athletes / heavy sweating | Increased requirement |
Dietary Sources of Potassium
A. Plant Sources (Richest)
-
Bananas
-
Oranges
-
Citrus fruits
-
Tomatoes
-
Potatoes
-
Green leafy vegetables
-
Pulses and legumes
B. Animal Sources
-
Meat
-
Fish
-
Milk and milk products
Absorption of Potassium
-
Potassium is rapidly and almost completely absorbed
-
Absorption occurs mainly in the small intestine
-
Absorption efficiency is >90%
-
Excess potassium is excreted mainly by the kidneys
Factors Promoting Potassium Absorption / Retention
-
Adequate dietary intake
-
Normal renal function
-
Insulin (drives potassium into cells)
-
Aldosterone (renal regulation)
Factors Inhibiting Potassium Absorption / Causing Loss
-
Chronic diarrhea and vomiting
-
Excessive sweating
-
Diuretic therapy (loop and thiazide diuretics)
-
Hyperaldosteronism
-
Laxative abuse
Disease States Related to Potassium
A. Hypokalemia
(Serum K⁺ < 3.5 mEq/L)
Causes:
-
Prolonged vomiting or diarrhea
-
Diuretic use
-
Hyperaldosteronism
-
Inadequate intake
Clinical Features:
-
Muscle weakness
-
Fatigue
-
Paralysis (severe cases)
-
Cardiac arrhythmias
-
Flattened T-wave on ECG
B. Hyperkalemia
(Serum K⁺ > 5.0 mEq/L)
Causes:
-
Renal failure
-
Potassium-sparing diuretics
-
Excess potassium intake
-
Acidosis
-
Tissue breakdown (burns, hemolysis)
Clinical Features:
-
Muscle weakness
-
Paresthesia
-
Life-threatening cardiac arrhythmias
-
Tall peaked T-waves on ECG
-
Cardiac arrest (severe cases)
C. Cardiac Arrhythmias
-
Both hypo- and hyperkalemia severely affect cardiac conduction
-
Potassium imbalance is a medical emergency
Hormonal Regulation of Potassium Balance
-
Aldosterone: Increases potassium excretion
-
Insulin: Promotes potassium entry into cells
-
Catecholamines: Shift potassium intracellularly
Chloride
Chloride is the major extracellular anion and an essential macromineral. It usually exists in association with sodium and potassium and plays a vital role in maintenance of osmotic pressure, acid–base balance, gastric acid formation, and electrolyte equilibrium.
Biochemical Functions of Chloride
A. Maintenance of Osmotic Pressure
-
Chloride, along with sodium, regulates extracellular fluid volume
-
Maintains electrical neutrality in body fluids
B. Acid–Base Balance
-
Involved in chloride–bicarbonate exchange (Hamburger phenomenon)
-
Helps maintain blood pH
-
Regulates bicarbonate concentration in plasma
C. Formation of Gastric Hydrochloric Acid
-
Chloride combines with hydrogen ions to form HCl in gastric parietal cells
-
Essential for:
-
Activation of pepsinogen
-
Protein digestion
-
Absorption of iron and vitamin B₁₂
-
D. Nerve and Muscle Function
-
Helps maintain membrane potential
-
Assists in neuromuscular excitability
-
Participates in inhibitory neurotransmission
E. Transport of Carbon Dioxide
-
Chloride shift facilitates CO₂ transport from tissues to lungs
-
Essential for efficient gas exchange
Dietary Requirements of Chloride
| Age / Condition | Requirement |
|---|---|
| Adults | 2300 mg/day |
| Pregnant women | 2300 mg/day |
| Lactating mothers | 2300 mg/day |
| Upper safe limit | 3600 mg/day |
Chloride requirement is usually met through common salt (NaCl) intake.
Dietary Sources of Chloride
A. Major Sources
-
Common salt (NaCl)
-
Processed and preserved foods
-
Pickles
-
Sauces and condiments
B. Natural Sources
-
Milk
-
Meat
-
Eggs
-
Vegetables
Absorption of Chloride
-
Chloride is rapidly and efficiently absorbed
-
Absorption occurs mainly in the small intestine
-
Absorption efficiency is >90%
-
Follows sodium absorption via electroneutral transport
Factors Promoting Chloride Absorption / Retention
-
Adequate sodium intake
-
Normal intestinal mucosa
-
Aldosterone (renal retention)
-
Acidic gastric environment
Factors Inhibiting Chloride Absorption / Causing Loss
-
Prolonged vomiting (loss of gastric HCl)
-
Chronic diarrhea
-
Excessive sweating
-
Diuretic therapy
-
Adrenal insufficiency
Disease States Related to Chloride
A. Hypochloremia
(Serum Cl⁻ < 98 mEq/L)
Causes:
-
Prolonged vomiting
-
Gastric suction
-
Diuretic use
-
Metabolic alkalosis
Clinical Features:
-
Muscle cramps
-
Weakness
-
Shallow breathing
-
Metabolic alkalosis
B. Hyperchloremia
(Serum Cl⁻ > 106 mEq/L)
Causes:
-
Dehydration
-
Excess saline infusion
-
Renal tubular acidosis
-
Metabolic acidosis
Clinical Features:
-
Deep and rapid breathing
-
Weakness
-
Confusion
-
Acid–base imbalance
C. Acid–Base Disorders
-
Chloride imbalance is commonly associated with:
-
Metabolic acidosis
-
Metabolic alkalosis
-
Microminerals
Iron
Iron is an essential trace element required primarily for oxygen transport, cellular respiration, and enzymatic reactions. The total body iron content is about 3–5 g, most of which is present in hemoglobin, with the remainder stored in ferritin and hemosiderin or present in myoglobin and enzymes.
Biochemical Functions of Iron
A. Oxygen Transport
-
Iron is a constituent of hemoglobin
-
Responsible for reversible binding and transport of oxygen
B. Oxygen Storage
-
Present in myoglobin in muscle
-
Facilitates oxygen availability during muscle contraction
C. Role in Cellular Respiration
-
Component of cytochromes (a, b, c)
-
Essential for electron transport chain and ATP production
D. Enzyme Functions
-
Cofactor for several enzymes:
-
Catalase
-
Peroxidase
-
Cytochrome oxidase
-
-
Required for detoxification reactions
E. Hemopoiesis
-
Essential for RBC formation and maturation
-
Supports normal erythropoiesis
F. Immune Function
-
Required for normal immune response
-
Involved in proliferation of immune cells
Dietary Requirements of Iron
| Age / Condition | Requirement (mg/day) |
|---|---|
| Adult males | 8–10 mg |
| Adult females | 18 mg |
| Pregnant women | 27 mg |
| Lactating mothers | 9–10 mg |
| Children | 7–10 mg |
Dietary Sources of Iron
A. Heme Iron (Better Absorbed)
-
Liver
-
Red meat
-
Fish
-
Poultry
B. Non-Heme Iron
-
Green leafy vegetables
-
Pulses and legumes
-
Whole grains
-
Nuts and seeds
-
Dates and jaggery
Absorption of Iron
-
Absorbed mainly in the duodenum and upper jejunum
-
Heme iron is absorbed more efficiently
-
Non-heme iron must be reduced from Fe³⁺ to Fe²⁺
-
Absorption is regulated by body iron stores
Factors Promoting Iron Absorption
-
Vitamin C (ascorbic acid)
-
Gastric acid (low pH)
-
Presence of meat (meat factor)
-
Increased erythropoiesis
-
Iron deficiency state
Factors Inhibiting Iron Absorption
-
Phytates (cereals)
-
Oxalates
-
Tannins (tea, coffee)
-
Excess calcium
-
Antacids
-
Chronic inflammation
Transport and Storage of Iron
Transport
-
Iron is transported in plasma bound to transferrin
Storage
-
Stored as:
-
Ferritin (water-soluble)
-
Hemosiderin (insoluble)
-
Disease States Related to Iron
A. Iron Deficiency Anemia
(Most common nutritional deficiency)
Causes:
-
Poor dietary intake
-
Chronic blood loss
-
Increased requirement (pregnancy)
-
Malabsorption
Clinical Features:
-
Pallor
-
Fatigue
-
Breathlessness
-
Glossitis
-
Koilonychia (spoon nails)
B. Anemia of Chronic Disease
-
Iron trapped in macrophages
-
Normal or increased ferritin
-
Reduced serum iron
C. Hemochromatosis
(Iron overload disorder)
Causes:
-
Genetic (primary)
-
Repeated blood transfusions (secondary)
Clinical Features:
-
Liver cirrhosis
-
Diabetes mellitus
-
Skin pigmentation
-
Cardiomyopathy
D. Hemosiderosis
-
Excess iron deposition in tissues
-
Less tissue damage compared to hemochromatosis
Zinc
Zinc is an essential trace element involved in a wide range of biochemical, catalytic, structural, and regulatory functions. The total body zinc content is about 2–3 g, with highest concentrations in muscle, bone, skin, liver, pancreas, and prostate. Zinc is crucial for growth, immune function, wound healing, and gene expression.
Biochemical Functions of Zinc
A. Enzyme Cofactor
-
Zinc is a cofactor for over 300 enzymes
-
Important zinc-dependent enzymes include:
-
Carbonic anhydrase
-
Alkaline phosphatase
-
Alcohol dehydrogenase
-
DNA and RNA polymerases
-
B. Role in Protein and Nucleic Acid Synthesis
-
Required for DNA replication and transcription
-
Essential for cell division and growth
C. Gene Expression
-
Structural component of zinc finger proteins
-
Regulates gene transcription and hormone receptor binding
D. Immune Function
-
Essential for T-cell maturation
-
Enhances cell-mediated immunity
-
Deficiency leads to recurrent infections
E. Wound Healing
-
Required for collagen synthesis
-
Promotes tissue repair and regeneration
F. Growth and Sexual Maturation
-
Required for normal growth in children
-
Essential for spermatogenesis and testosterone synthesis
G. Sensory Functions
-
Necessary for taste (gustation) and smell (olfaction)
-
Deficiency causes hypogeusia
Dietary Requirements of Zinc
| Age / Condition | Requirement (mg/day) |
|---|---|
| Adult males | 11 mg |
| Adult females | 8 mg |
| Pregnant women | 11–12 mg |
| Lactating mothers | 12–13 mg |
| Children | 5–10 mg |
Dietary Sources of Zinc
A. Animal Sources (Better Absorbed)
-
Meat
-
Liver
-
Fish
-
Eggs
-
Milk and milk products
B. Plant Sources
-
Whole grains
-
Pulses and legumes
-
Nuts and seeds
-
Green leafy vegetables
Absorption of Zinc
-
Absorbed mainly in the small intestine (jejunum)
-
Absorption occurs via:
-
Carrier-mediated transport
-
Passive diffusion
-
-
About 20–40% of dietary zinc is absorbed
-
Absorption is influenced by dietary composition
Factors Promoting Zinc Absorption
-
Animal protein (meat factor)
-
Adequate dietary intake
-
Low phytate content
-
Increased physiological demand (growth, pregnancy)
Factors Inhibiting Zinc Absorption
-
Phytates (whole grains, cereals)
-
High dietary fiber
-
Excess iron or calcium supplementation
-
Chronic diarrhea
-
Alcoholism
Transport and Storage of Zinc
Transport
-
Transported in plasma bound to:
-
Albumin
-
α-macroglobulin
-
Storage
-
No specific storage form
-
Distributed in tissues bound to proteins and enzymes
Disease States Related to Zinc
A. Zinc Deficiency
Causes:
-
Inadequate dietary intake
-
Malabsorption syndromes
-
Chronic diarrhea
-
Alcoholism
-
Long-term parenteral nutrition without zinc
Clinical Features:
-
Growth retardation
-
Delayed sexual maturation
-
Poor wound healing
-
Dermatitis
-
Alopecia
-
Loss of taste and smell
-
Recurrent infections
B. Acrodermatitis Enteropathica
-
Rare genetic disorder
-
Impaired zinc absorption
-
Presents with:
-
Severe dermatitis
-
Diarrhea
-
Alopecia
-
-
Treated with lifelong zinc supplementation
C. Zinc Toxicity
Causes:
-
Excess supplementation
-
Industrial exposure
Clinical Features:
-
Nausea and vomiting
-
Abdominal pain
-
Interference with copper absorption
-
Secondary copper deficiency anemia
Copper
Copper is an essential trace element required for iron metabolism, connective tissue formation, antioxidant defense, and nervous system function. The total body copper content is about 80–100 mg, with the highest concentrations in the liver, brain, heart, kidneys, and muscles.
Biochemical Functions of Copper
A. Iron Metabolism
-
Copper is essential for iron absorption and utilization
-
Component of ceruloplasmin, which oxidizes Fe²⁺ to Fe³⁺ for binding with transferrin
-
Copper deficiency can cause secondary iron deficiency anemia
B. Enzyme Cofactor
Copper is a cofactor for several important enzymes:
-
Cytochrome c oxidase – cellular respiration
-
Superoxide dismutase (Cu-Zn SOD) – antioxidant defense
-
Tyrosinase – melanin synthesis
-
Lysyl oxidase – collagen and elastin cross-linking
-
Dopamine β-hydroxylase – catecholamine synthesis
C. Connective Tissue Formation
-
Required for cross-linking of collagen and elastin
-
Essential for bone strength, vascular integrity, and skin health
D. Antioxidant Defense
-
Copper-containing enzymes protect cells from oxidative damage
-
Works synergistically with zinc
E. Nervous System Function
-
Required for myelination and neurotransmitter synthesis
-
Essential for normal brain development
F. Pigmentation
-
Necessary for synthesis of melanin
-
Deficiency leads to hypopigmentation
Dietary Requirements of Copper
| Age / Condition | Requirement (mg/day) |
|---|---|
| Adults | 0.9 mg |
| Pregnant women | 1.0 mg |
| Lactating mothers | 1.3 mg |
| Children | 0.3–0.9 mg |
Dietary Sources of Copper
A. Rich Sources
-
Liver
-
Shellfish
-
Nuts and seeds
-
Cocoa
-
Whole grains
B. Other Sources
-
Legumes
-
Green leafy vegetables
-
Drinking water (from copper pipes)
Absorption of Copper
-
Absorbed mainly in the stomach and small intestine
-
About 30–50% of dietary copper is absorbed
-
Absorption decreases with high intake
Factors Promoting Copper Absorption
-
Adequate dietary intake
-
Animal protein
-
Acidic gastric pH
-
Low body copper stores
Factors Inhibiting Copper Absorption
-
Excess zinc intake
-
High iron intake
-
Phytates
-
Chronic diarrhea
-
Malabsorption syndromes
Transport and Storage of Copper
Transport
-
Transported in plasma bound to:
-
Albumin (initial transport)
-
Ceruloplasmin (major circulating form)
-
Storage
-
Stored mainly in the liver
-
Excess copper excreted in bile
Disease States Related to Copper
A. Copper Deficiency
Causes:
-
Malnutrition
-
Malabsorption
-
Excess zinc intake
-
Long-term parenteral nutrition
Clinical Features:
-
Hypochromic anemia
-
Neutropenia
-
Bone abnormalities
-
Hypopigmentation
-
Neurological symptoms
B. Menkes Disease
-
X-linked genetic disorder
-
Defective copper absorption and transport
-
Leads to:
-
Kinky hair
-
Growth failure
-
Neurological deterioration
-
-
Usually fatal in early childhood
C. Wilson’s Disease
-
Autosomal recessive disorder
-
Excess copper accumulation in tissues
-
Defective biliary excretion of copper
Clinical Features:
-
Liver cirrhosis
-
Neurological symptoms
-
Kayser–Fleischer rings in eyes
-
Psychiatric manifestations
D. Copper Toxicity
Causes:
-
Excess supplementation
-
Industrial exposure
Clinical Features:
-
Gastrointestinal distress
-
Liver damage
-
Hemolysis (severe cases)
Manganese
Manganese is an essential trace required for enzyme activation, bone formation, metabolism of carbohydrates, amino acids, and cholesterol, and antioxidant defense. The total body manganese content is small, with highest concentrations in the liver, pancreas, kidneys, and bones.
Biochemical Functions of Manganese
A. Enzyme Cofactor
Manganese acts as a cofactor for several enzymes, including:
-
Pyruvate carboxylase – gluconeogenesis
-
Arginase – urea cycle
-
Superoxide dismutase (Mn-SOD) – mitochondrial antioxidant defense
-
Glycosyltransferases – proteoglycan synthesis
B. Carbohydrate, Lipid, and Protein Metabolism
-
Required for enzymes involved in:
-
Glucose metabolism
-
Fatty acid synthesis
-
Amino acid metabolism
-
C. Bone Formation and Growth
-
Essential for osteoblast function
-
Involved in bone matrix formation
-
Required for normal growth and skeletal development
D. Antioxidant Defense
-
Component of mitochondrial superoxide dismutase (Mn-SOD)
-
Protects cells from oxidative stress
E. Reproductive Function
-
Necessary for normal reproductive hormone synthesis
-
Supports fertility and normal sexual development
F. Nervous System Function
-
Required for normal brain function
-
Involved in neurotransmitter metabolism
Dietary Requirements of Manganese
| Age / Condition | Requirement (mg/day) |
|---|---|
| Adult males | 2.3 mg |
| Adult females | 1.8 mg |
| Pregnant women | 2.0 mg |
| Lactating mothers | 2.6 mg |
| Children | 1.2–1.5 mg |
Dietary Sources of Manganese
A. Rich Sources
-
Whole grains
-
Nuts and seeds
-
Tea
-
Legumes
-
Green leafy vegetables
B. Other Sources
-
Fruits
-
Spices
-
Drinking water (trace amounts)
Absorption of Manganese
-
Absorbed mainly in the small intestine
-
Absorption efficiency is low (3–5%)
-
Excess manganese is excreted mainly through bile
Factors Promoting Manganese Absorption
-
Low dietary manganese intake
-
Iron deficiency (increases Mn absorption)
-
Normal intestinal function
Factors Inhibiting Manganese Absorption
-
High dietary iron
-
High calcium intake
-
Phytates and fiber
-
Malabsorption syndromes
Transport and Storage of Manganese
Transport
-
Transported in plasma bound to:
-
Transferrin
-
Albumin
-
Storage
-
Stored mainly in the liver and bones
Disease States Related to Manganese
A. Manganese Deficiency
(Rare)
Causes:
-
Poor dietary intake
-
Long-term parenteral nutrition without Mn
Clinical Features:
-
Impaired growth
-
Skeletal abnormalities
-
Altered glucose metabolism
-
Reduced fertility
B. Manganese Toxicity
(Common in occupational exposure)
Causes:
-
Industrial inhalation (mining, welding)
-
Excess supplementation
Clinical Features:
-
Neurological symptoms (manganism)
-
Parkinson-like features
-
Tremors
-
Behavioral changes
C. Neurological Disorders
-
Excess manganese accumulates in basal ganglia
-
Leads to irreversible neurological damage
Selenium
Selenium is an essential trace element required in very small amounts for antioxidant defense, thyroid hormone metabolism, immune function, and reproductive health. Selenium is biologically active as selenoproteins, in which selenium is present as the amino acid selenocysteine.
Biochemical Functions of Selenium
A. Antioxidant Defense
-
Selenium is a component of glutathione peroxidase
-
Protects cells from oxidative damage by reducing hydrogen peroxide and lipid peroxides
-
Works synergistically with vitamin E
B. Thyroid Hormone Metabolism
-
Component of iodothyronine deiodinases
-
Converts thyroxine (T₄) to triiodothyronine (T₃)
-
Protects thyroid gland from oxidative stress
C. Immune Function
-
Enhances cell-mediated and humoral immunity
-
Supports proliferation of T lymphocytes
-
Improves resistance to infections
D. Reproductive Function
-
Required for normal spermatogenesis
-
Maintains sperm motility
-
Important for fertility
E. Cardiovascular Protection
-
Prevents oxidative damage to lipoproteins
-
May reduce risk of atherosclerosis
Dietary Requirements of Selenium
| Age / Condition | Requirement (µg/day) |
|---|---|
| Adults | 55 µg |
| Pregnant women | 60 µg |
| Lactating mothers | 70 µg |
| Children | 20–40 µg |
Dietary Sources of Selenium
A. Rich Sources
-
Brazil nuts (very rich source)
-
Seafood
-
Organ meats
-
Eggs
B. Other Sources
-
Cereals and grains
-
Milk and dairy products
-
Vegetables (content depends on soil selenium)
Absorption of Selenium
-
Selenium is efficiently absorbed (≈80–90%)
-
Absorbed mainly in the small intestine
-
Organic forms (selenium in food proteins) are better absorbed than inorganic forms
Factors Promoting Selenium Absorption
-
Adequate protein intake
-
Organic selenium forms (selenocysteine, selenomethionine)
-
Normal intestinal function
Factors Inhibiting Selenium Absorption
-
Heavy metals (mercury, cadmium)
-
Poor intestinal absorption
-
High sulfur intake
-
Chronic diarrhea
Transport and Storage of Selenium
Transport
-
Transported in plasma bound to selenoprotein P
Storage
-
Stored in:
-
Liver
-
Kidney
-
Muscle
-
-
No large storage pool; regular intake required
Disease States Related to Selenium
A. Selenium Deficiency
Causes:
-
Low selenium content in soil
-
Malnutrition
-
Long-term parenteral nutrition
Clinical Features:
-
Keshan disease (cardiomyopathy)
-
Kashin–Beck disease (osteoarthropathy)
-
Muscle weakness
-
Increased susceptibility to infections
-
Hypothyroidism-like features
B. Keshan Disease
-
Endemic cardiomyopathy
-
Seen in selenium-deficient regions
-
Affects children and women of childbearing age
C. Selenium Toxicity (Selenosis)
Causes:
-
Excess supplementation
-
High intake from selenium-rich foods
Clinical Features:
-
Garlic-like breath odor
-
Hair loss
-
Brittle nails
-
Gastrointestinal upset
-
Neurological abnormalities
Iodine
Iodine is an essential trace (micromineral) required for the synthesis of thyroid hormones, which regulate basal metabolic rate, growth, development, and energy metabolism. The total body iodine content is about 15–20 mg, with nearly 70–80% stored in the thyroid gland.
Biochemical Functions of Iodine
A. Thyroid Hormone Synthesis
-
Iodine is an essential component of:
-
Thyroxine (T₄)
-
Triiodothyronine (T₃)
-
-
Required for iodination of tyrosine residues in thyroglobulin
B. Regulation of Basal Metabolic Rate (BMR)
-
Thyroid hormones regulate:
-
Energy expenditure
-
Oxygen consumption
-
Heat production
-
C. Growth and Development
-
Essential for normal physical growth
-
Critical for brain development, especially during fetal life and infancy
D. Nervous System Development
-
Required for myelination and neuronal differentiation
-
Deficiency during pregnancy leads to irreversible brain damage
E. Protein, Fat, and Carbohydrate Metabolism
-
Thyroid hormones influence:
-
Protein synthesis
-
Lipolysis
-
Glucose metabolism
-
Dietary Requirements of Iodine
| Age / Condition | Requirement (µg/day) |
|---|---|
| Adults | 150 µg |
| Pregnant women | 220 µg |
| Lactating mothers | 290 µg |
| Children | 90–120 µg |
Dietary Sources of Iodine
A. Rich Sources
-
Iodized salt (major source)
-
Sea fish
-
Seaweed
B. Other Sources
-
Milk and dairy products
-
Eggs
-
Meat
-
Cereals and vegetables (iodine content depends on soil)
Absorption of Iodine
-
Iodine is absorbed in the small intestine
-
Absorption efficiency is >90%
-
Absorbed iodine circulates as iodide
-
Taken up by the thyroid via sodium–iodide symporter (NIS)
Factors Promoting Iodine Absorption and Utilization
-
Adequate dietary intake
-
Normal gastrointestinal function
-
Proper thyroid function
-
Adequate selenium status (for hormone activation)
Factors Inhibiting Iodine Absorption / Utilization
-
Goitrogens (thiocyanates in cabbage, cauliflower)
-
Excess intake of goitrogenic foods
-
Iodine deficiency
-
Certain drugs (amiodarone, lithium)
-
Smoking (thiocyanates)
Transport and Storage of Iodine
Transport
-
Transported in blood as iodide
Storage
-
Stored mainly in the thyroid gland
-
Incorporated into thyroglobulin
Disease States Related to Iodine
A. Iodine Deficiency Disorders (IDD)
Includes:
-
Endemic goiter
-
Hypothyroidism
-
Cretinism
-
Mental retardation
B. Goiter
-
Enlargement of thyroid gland
-
Due to inadequate iodine intake
-
Caused by increased TSH stimulation
C. Cretinism
-
Severe iodine deficiency during fetal life
-
Characterized by:
-
Mental retardation
-
Dwarfism
-
Deaf-mutism
-
Delayed sexual development
-
D. Hypothyroidism
-
Low levels of thyroid hormones
-
Features:
-
Weight gain
-
Cold intolerance
-
Lethargy
-
Bradycardia
-
E. Hyperthyroidism (Excess Iodine)
-
Excess iodine intake can trigger:
-
Iodine-induced hyperthyroidism
-
-
Seen in susceptible individuals
Prevention of Iodine Deficiency
-
Universal salt iodization
-
Health education
-
Monitoring iodine content in salt
MCQs
1. Minerals are best defined as:
A. Organic nutrients
B. Energy-yielding substances
C. Inorganic nutrients required for body functions
D. Vitamins
Answer: C
2. Minerals do NOT provide:
A. Structural support
B. Enzyme activation
C. Energy
D. Electrolyte balance
Answer: C
3. Minerals required in amounts greater than 100 mg/day are called:
A. Trace minerals
B. Ultratrace elements
C. Macrominerals
D. Micronutrients
Answer: C
4. Which of the following is a macromineral?
A. Zinc
B. Iron
C. Calcium
D. Selenium
Answer: C
5. Which mineral is the major extracellular cation?
A. Potassium
B. Calcium
C. Sodium
D. Magnesium
Answer: C
6. Which is the major intracellular cation?
A. Sodium
B. Potassium
C. Calcium
D. Chloride
Answer: B
7. The major extracellular anion is:
A. Bicarbonate
B. Phosphate
C. Chloride
D. Sulfate
Answer: C
8. About 99% of calcium is found in:
A. Blood
B. Muscles
C. Bones and teeth
D. Liver
Answer: C
9. Calcium is required for all EXCEPT:
A. Muscle contraction
B. Blood coagulation
C. Nerve transmission
D. Oxygen transport
Answer: D
10. Vitamin D primarily helps in absorption of:
A. Iron
B. Zinc
C. Calcium
D. Sodium
Answer: C
11. Which factor inhibits calcium absorption?
A. Vitamin D
B. Lactose
C. Phytates
D. Growth hormone
Answer: C
12. The second most abundant mineral in the body is:
A. Magnesium
B. Phosphorus
C. Sodium
D. Potassium
Answer: B
13. Phosphorus is an essential component of:
A. Hemoglobin
B. ATP
C. Vitamin D
D. Insulin
Answer: B
14. 2,3-BPG in RBCs contains:
A. Calcium
B. Iron
C. Phosphorus
D. Magnesium
Answer: C
15. Magnesium acts mainly as:
A. Structural mineral
B. Enzyme cofactor
C. Hormone
D. Buffer
Answer: B
16. Magnesium deficiency commonly causes:
A. Hypertension
B. Tetany
C. Muscle relaxation
D. Hypercalcemia
Answer: B
17. Iron is mainly absorbed in:
A. Ileum
B. Colon
C. Duodenum
D. Stomach
Answer: C
18. The transport protein for iron is:
A. Albumin
B. Ferritin
C. Transferrin
D. Ceruloplasmin
Answer: C
19. Storage form of iron is:
A. Hemoglobin
B. Transferrin
C. Ferritin
D. Cytochrome
Answer: C
20. Iron deficiency causes:
A. Megaloblastic anemia
B. Hemolytic anemia
C. Microcytic hypochromic anemia
D. Aplastic anemia
Answer: C
21. Zinc is required for:
A. Oxygen transport
B. Enzyme activation
C. Bone mineralization only
D. Gastric acid formation
Answer: B
22. Zinc deficiency commonly causes:
A. Night blindness
B. Loss of taste
C. Goiter
D. Anemia
Answer: B
23. Acrodermatitis enteropathica is due to deficiency of:
A. Copper
B. Zinc
C. Iron
D. Selenium
Answer: B
24. Copper is essential for:
A. Thyroid hormone synthesis
B. Iron metabolism
C. Sodium balance
D. Calcium absorption
Answer: B
25. Ceruloplasmin is associated with:
A. Iron transport
B. Copper metabolism
C. Zinc storage
D. Calcium regulation
Answer: B
26. Wilson’s disease is due to excess:
A. Iron
B. Zinc
C. Copper
D. Selenium
Answer: C
27. Manganese is a component of:
A. Hemoglobin
B. Glutathione peroxidase
C. Mn-superoxide dismutase
D. Cytochrome oxidase
Answer: C
28. Selenium acts mainly as:
A. Structural mineral
B. Antioxidant
C. Electrolyte
D. Hormone
Answer: B
29. Selenium is a component of:
A. Catalase
B. Glutathione peroxidase
C. Carbonic anhydrase
D. Tyrosinase
Answer: B
30. Keshan disease is associated with deficiency of:
A. Iodine
B. Selenium
C. Iron
D. Zinc
Answer: B
31. Iodine is essential for synthesis of:
A. Insulin
B. Cortisol
C. Thyroid hormones
D. Growth hormone
Answer: C
32. Major storage site of iodine is:
A. Liver
B. Kidney
C. Thyroid gland
D. Muscle
Answer: C
33. Iodine deficiency in children leads to:
A. Scurvy
B. Rickets
C. Cretinism
D. Pellagra
Answer: C
34. Goiter is caused by:
A. Excess iodine
B. Iodine deficiency
C. Selenium excess
D. Zinc deficiency
Answer: B
35. Sodium and potassium balance is mainly regulated by:
A. Insulin
B. Thyroxine
C. Aldosterone
D. Parathyroid hormone
Answer: C
36. Chloride is required for formation of:
A. Pepsin
B. Bile salts
C. Hydrochloric acid
D. Trypsin
Answer: C
37. Chloride shift occurs in:
A. Liver
B. Kidneys
C. RBCs
D. Intestine
Answer: C
38. Hypokalemia primarily affects:
A. Liver
B. Bone
C. Heart rhythm
D. Vision
Answer: C
39. Hyperkalemia may cause:
A. Hypertension
B. Cardiac arrest
C. Osteoporosis
D. Anemia
Answer: B
40. Sodium excess is commonly associated with:
A. Hypotension
B. Hypertension
C. Hypoglycemia
D. Anemia
Answer: B
41. Phytates reduce absorption of:
A. Sodium
B. Potassium
C. Iron and zinc
D. Chloride
Answer: C
42. Oxalates inhibit absorption of:
A. Iron
B. Calcium
C. Sodium
D. Selenium
Answer: B
43. Which mineral is required for blood coagulation?
A. Iron
B. Calcium
C. Zinc
D. Copper
Answer: B
44. The phosphate buffer system helps maintain:
A. Osmotic pressure
B. Blood glucose
C. Acid–base balance
D. Oxygen transport
Answer: C
45. Most abundant mineral in the body is:
A. Phosphorus
B. Calcium
C. Magnesium
D. Sodium
Answer: B
46. Minerals are absorbed mainly from:
A. Stomach
B. Colon
C. Small intestine
D. Rectum
Answer: C
47. Which mineral deficiency causes osteoporosis?
A. Iron
B. Zinc
C. Calcium
D. Sodium
Answer: C
48. Which mineral interacts with insulin action?
A. Chromium
B. Sodium
C. Chloride
D. Selenium
Answer: A
49. Ultratrace elements include:
A. Calcium
B. Zinc
C. Nickel
D. Iron
Answer: C
50. The best source of iodine in diet is:
A. Milk
B. Green vegetables
C. Iodized salt
D. Fruits
Answer: C
