Introduction
-
Plasma lipids include cholesterol, triglycerides, phospholipids, and free fatty acids, which are essential for energy metabolism, membrane structure, and hormone synthesis.
-
Because lipids are insoluble in water, they circulate in blood as lipoproteins, composed of lipids and specific apolipoproteins.
-
Disorders of plasma lipids and lipoproteins arise due to abnormal synthesis, transport, or clearance of lipoproteins.
-
These disorders lead to either decreased lipid levels (hypolipoproteinemia) or increased lipid levels (hyperlipoproteinemia / dyslipidemia).
-
Lipoprotein disorders may be primary (genetic) or secondary (acquired), with secondary causes being more common in clinical practice.
-
Dyslipidemias are major biochemical risk factors for atherosclerosis, cardiovascular disease, metabolic syndrome, and diabetes mellitus.
-
Altered lipid metabolism also affects oral and periodontal health, influencing wound healing, alveolar bone health, and outcomes of dental procedures.
-
A sound understanding of these disorders helps students correlate biochemical mechanisms with clinical and dental manifestations.
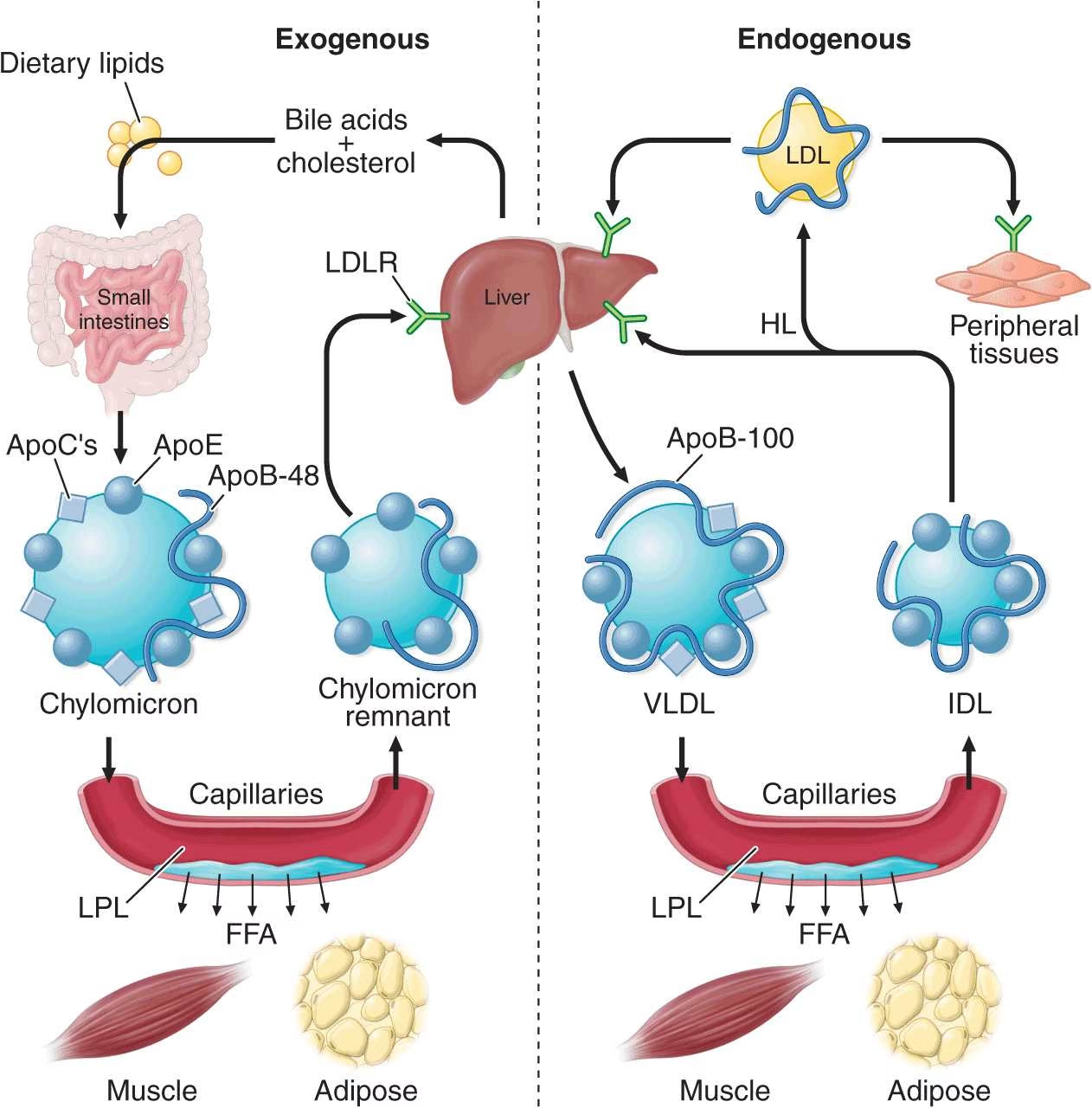
Normal Plasma Lipids
Major plasma lipids include:
-
Triglycerides (TAG)
-
Cholesterol
-
Cholesteryl esters
-
Phospholipids
-
Free fatty acids
Normal lipid transport occurs via:
-
Chylomicrons
-
VLDL
-
IDL
-
LDL
-
HDL
Lipoproteins – Composition & Function
| Lipoprotein | Major Lipid | Function |
|---|---|---|
| Chylomicrons | Triglycerides (dietary) | Transport dietary fat |
| VLDL | Triglycerides (endogenous) | Transport liver-derived TAG |
| LDL | Cholesterol | Deliver cholesterol to tissues |
| HDL | Cholesterol | Reverse cholesterol transport |
Classification of Disorders of Plasma Lipids
Broad Classification
-
Hypolipoproteinemia – ↓ plasma lipoproteins
-
Hyperlipoproteinemia – ↑ plasma lipoproteins
Each is further divided into:
-
Primary (genetic)
-
Secondary (acquired)
I. Hypolipoproteinemia
(Decreased plasma lipids/lipoproteins)
Hypolipoproteinemia is characterized by abnormally low levels of LDL, HDL, or both, leading to impaired lipid transport and deficiency of fat-soluble vitamins.
A. Primary Hypolipoproteinemia (Genetic)
1. Disorders with Low LDL Cholesterol
| Disorder | Biochemical Defect | Key Features |
|---|---|---|
| Abetalipoproteinemia | Absence of Apo-B | No chylomicrons, VLDL, LDL |
| Hypobetalipoproteinemia | Apo-B mutation | Low LDL |
| PCSK9 deficiency | ↑ LDL receptor activity | Markedly low LDL |
| Chylomicron retention disease | Defective chylomicron secretion | Fat malabsorption |
| Familial combined hypolipidemia | Combined Apo defects | Low LDL & HDL |
2. Disorders with Low HDL Cholesterol
| Disorder | Defect | Characteristic Feature |
|---|---|---|
| LCAT deficiency | Defective cholesterol esterification | Corneal opacity |
| Apo A-I deficiency | Impaired HDL synthesis | Low HDL |
| Familial hypoalphalipoproteinemia | Reduced HDL | Atherosclerosis risk |
| Tangier disease | HDL almost absent | Orange tonsils |
| Fish-eye disease | Partial LCAT deficiency | Corneal opacity |
B. Secondary Hypolipoproteinemia (Acquired)
Common Causes
-
Anemia
-
Chronic inflammation
-
Chronic liver disease
-
Hyperthyroidism
-
Chronic infections
-
Malabsorption syndromes
-
Malignancy
-
Critical illness
Mechanism: Reduced hepatic synthesis or increased catabolism of lipoproteins.
II. Hyperlipoproteinemia
(Increased plasma lipids/lipoproteins)
Hyperlipoproteinemia refers to elevated cholesterol, triglycerides, or both, due to impaired clearance or overproduction of lipoproteins.
A. Primary Hyperlipoproteinemia (Genetic)
Fredrickson Classification
| Type | Lipoprotein Increased | Lipid Increased | Key Feature |
|---|---|---|---|
| Type I | Chylomicrons | Triglycerides | Pancreatitis |
| Type IIa | LDL | Cholesterol | Severe atherosclerosis |
| Type IIb | LDL + VLDL | Cholesterol + TAG | Most common |
| Type III | IDL | Chol + TAG | Palmar xanthomas |
| Type IV | VLDL | Triglycerides | Diabetes, obesity |
| Type V | VLDL + Chylomicrons | Triglycerides | Pancreatitis |
Important Primary Disorders
Type IIa – Familial Hypercholesterolemia
-
Defect: LDL receptor mutation
-
Biochemistry: ↑ LDL-cholesterol
-
Clinical: Tendon xanthomas, premature CAD
Type IIb – Familial Combined Hyperlipidemia
-
Most common genetic dyslipidemia
-
↑ LDL + VLDL
-
Strongly associated with cardiovascular disease
B. Secondary Hyperlipoproteinemia
Causes
| Condition | Lipid Pattern |
|---|---|
| Diabetes mellitus | ↑ TAG, ↓ HDL |
| Obesity | ↑ VLDL, ↑ TAG |
| Alcoholism | ↑ TAG |
| Hypothyroidism | ↑ LDL |
| Nephrotic syndrome | ↑ LDL |
| Liver disease | Mixed dyslipidemia |
| Drugs (steroids, OCPs) | ↑ TAG & cholesterol |
| Smoking | ↓ HDL |
Pathophysiology of Atherosclerosis
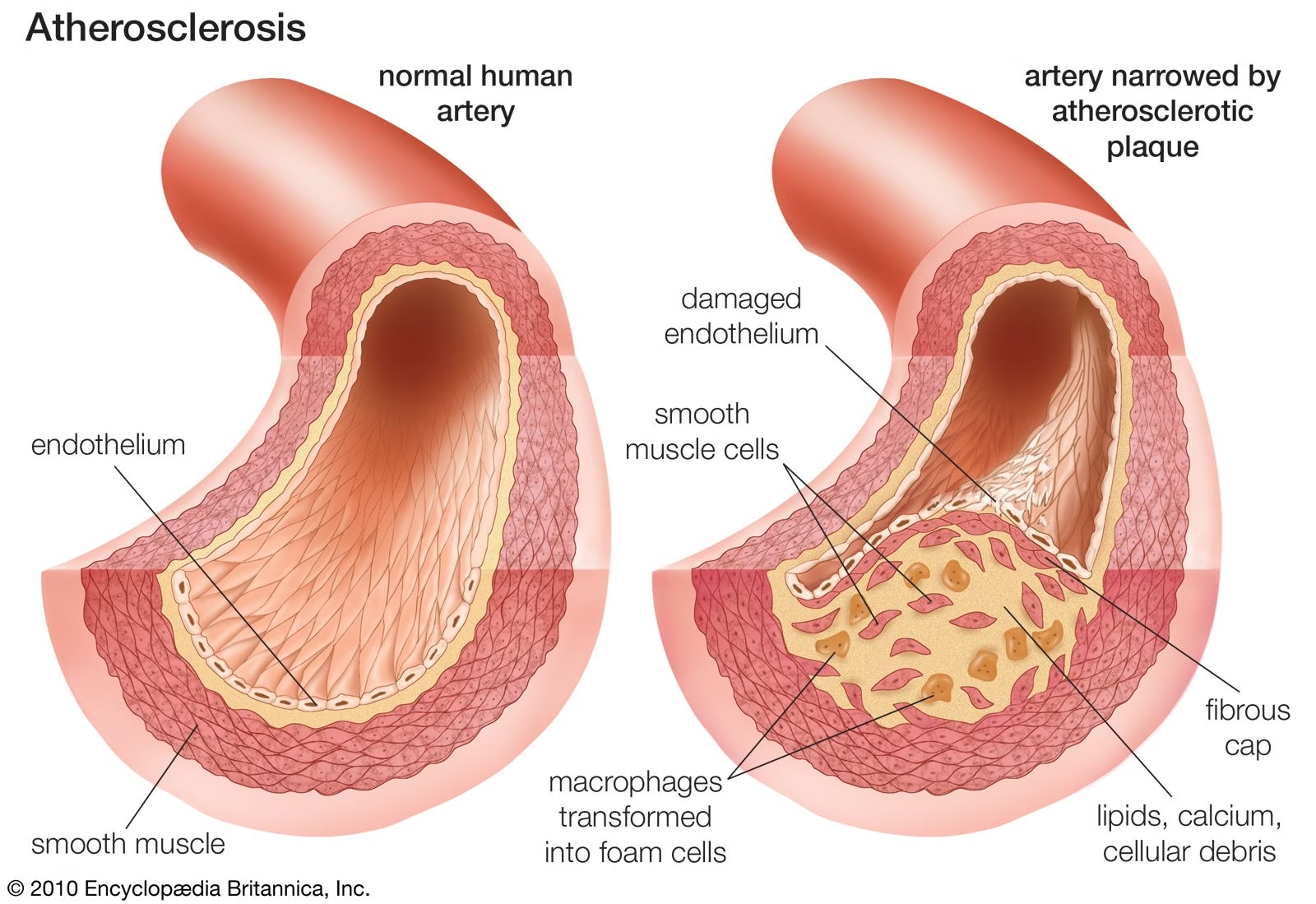
Step 1: Endothelial Dysfunction (Initiating Event)
-
Normal endothelium maintains vascular tone and antithrombotic state.
-
Risk factors cause endothelial injury:
-
Hyperlipidemia (↑ LDL)
-
Smoking
-
Diabetes mellitus
-
Hypertension
-
-
Endothelial damage leads to:
-
↑ Vascular permeability to lipoproteins
-
↑ Adhesion of monocytes
-
Step 2: Entry and Oxidation of LDL
-
LDL particles enter the subendothelial space (intima).
-
LDL undergoes oxidation by:
-
Reactive oxygen species (ROS)
-
Endothelial and macrophage enzymes
-
-
Oxidized LDL (oxLDL) is highly atherogenic.
Biochemical importance: Oxidized LDL is not recognized by normal LDL receptors.
Step 3: Monocyte Migration and Foam Cell Formation
-
Monocytes migrate into the intima and differentiate into macrophages.
-
Macrophages engulf oxidized LDL via scavenger receptors.
-
Lipid-laden macrophages are called foam cells.
Accumulation of foam cells forms fatty streaks (earliest lesion).
Step 4: Smooth Muscle Cell Proliferation
-
Growth factors released by:
-
Activated macrophages
-
Damaged endothelium
-
-
Cause:
-
Migration of smooth muscle cells from media to intima
-
Proliferation of smooth muscle cells
-
-
Smooth muscle cells synthesize:
-
Collagen
-
Proteoglycans
-
Elastin
-
Step 5: Formation of Fibrous Atherosclerotic Plaque
-
Lipid core composed of:
-
Cholesterol
-
Cholesteryl esters
-
Dead foam cells
-
-
Covered by a fibrous cap (collagen-rich)
-
Results in:
-
Narrowing of arterial lumen
-
Reduced blood flow (ischemia)
-
Step 6: Plaque Complications
-
Plaque rupture or erosion exposes thrombogenic material
-
Leads to:
-
Platelet aggregation
-
Thrombus formation
-
-
Can cause:
-
Myocardial infarction
-
Stroke
-
Peripheral vascular disease
-
Role of Lipoproteins in Atherosclerosis
| Lipoprotein | Role |
|---|---|
| LDL | Highly atherogenic |
| Oxidized LDL | Triggers inflammation |
| HDL | Protective (reverse cholesterol transport) |
Biochemical Risk Factors
-
↑ LDL cholesterol
-
↓ HDL cholesterol
-
↑ Triglycerides
-
Oxidative stress
-
Chronic inflammation
Laboratory Evaluation of Lipid Disorders
1. Specimen and Pre-analytical Requirements
-
Sample: Venous blood (serum preferred)
-
Fasting state:
-
9–12 hours fasting required
-
Only water allowed
-
-
Reason for fasting:
-
To eliminate post-prandial lipemia
-
To obtain accurate triglyceride and VLDL values
-
2. Fasting Lipid Profile (Primary Test)
The fasting lipid profile is the first-line and most important investigation.
Parameters Measured
| Parameter | Normal Reference Range |
|---|---|
| Total cholesterol (TC) | < 200 mg/dL |
| Triglycerides (TG) | < 150 mg/dL |
| HDL-cholesterol | > 40 mg/dL (men) > 50 mg/dL (women) |
| LDL-cholesterol | < 100 mg/dL (optimal) |
| VLDL-cholesterol | 5–40 mg/dL |
Diagnostic Significance
-
↑ Total cholesterol → Hypercholesterolemia
-
↑ Triglycerides → Hypertriglyceridemia
-
↑ LDL-C → Increased atherogenic risk
-
↓ HDL-C → Loss of cardioprotective effect
3. Calculation of Lipoprotein Fractions
Friedewald Formula (Very Important for Exams)
-
VLDL-C = TG / 5
-
LDL-C = TC − (HDL-C + VLDL-C)
Limitations:
-
Not valid when:
-
TG > 400 mg/dL
-
Non-fasting sample
-
Type I hyperlipoproteinemia
-
4. Atherogenic Indices (Risk Assessment)
Atherogenic ratios give better cardiovascular risk prediction than individual lipid values.
| Index | Formula | Clinical Significance |
|---|---|---|
| TC / HDL | Total cholesterol ÷ HDL | Overall CV risk |
| LDL / HDL | LDL ÷ HDL | Strong predictor |
| TG / HDL | TG ÷ HDL | Insulin resistance |
| Non-HDL cholesterol | TC − HDL | Total atherogenic lipoproteins |
High-risk TC/HDL ratio: > 5
5. Apolipoprotein Estimation
| Apolipoprotein | Diagnostic Role |
|---|---|
| Apo-B | Reflects number of atherogenic particles (LDL, VLDL) |
| Apo-A1 | Major protein of HDL |
| Apo-B / Apo-A1 ratio | Best predictor of CV risk |
Especially useful when LDL-C is misleading (e.g., high TG states)
6. Lipoprotein
-
LDL-like particle containing apolipoprotein(a)
-
Genetically determined
-
Highly atherogenic and thrombogenic
-
Indicated in:
-
Premature cardiovascular disease
-
Strong family history of CAD
-
Normal lipid profile with high risk
-
7. Specialized Lipoprotein Tests
-
Lipoprotein electrophoresis
-
Identifies Fredrickson types
-
-
Ultracentrifugation
-
Gold standard for lipoprotein separation
-
-
Genetic testing
-
Familial hypercholesterolemia
-
Apo-E polymorphism
-
8. Evaluation of Secondary Causes
Whenever dyslipidemia is detected, secondary causes must be ruled out.
| Condition | Laboratory Tests |
|---|---|
| Diabetes mellitus | Fasting glucose, HbA1c |
| Hypothyroidism | TSH |
| Nephrotic syndrome | Serum albumin, urine protein |
| Liver disease | LFT (AST, ALT, ALP, bilirubin) |
| Renal disease | Urea, creatinine |
| Alcoholism | Liver enzymes, TG |
9. Typical Laboratory Patterns
| Disorder | Characteristic Findings |
|---|---|
| Familial hypercholesterolemia | ↑ LDL, normal TG |
| Type IV hyperlipoproteinemia | ↑ TG, ↑ VLDL |
| Type I & V | Very high TG |
| Tangier disease | Extremely ↓ HDL |
| Abetalipoproteinemia | Very low TC & TG |
Management of Lipid Disorders
Principles of Management
-
Identify whether dyslipidemia is primary (genetic) or secondary (acquired)
-
Assess overall cardiovascular risk
-
Correct modifiable risk factors
-
Combine lifestyle modification with pharmacotherapy when required
-
Monitor lipid levels and treatment response regularly
I. Lifestyle Modification (First-Line for All Patients)
1. Dietary Management
Goals: Reduce LDL and triglycerides, increase HDL
-
Reduce saturated fats and trans fats
-
Limit dietary cholesterol
-
Increase intake of:
-
Fruits and vegetables
-
Whole grains
-
Dietary fiber
-
-
Prefer:
-
Polyunsaturated and monounsaturated fats
-
-
Avoid:
-
Excess sugar
-
Refined carbohydrates
-
Alcohol (especially in hypertriglyceridemia)
-
Diet alone can reduce LDL by 10–15%
2. Physical Activity
-
Regular aerobic exercise (≥150 minutes/week)
-
Effects:
-
↑ HDL cholesterol
-
↓ Triglycerides
-
Improves insulin sensitivity
-
3. Weight Reduction
-
Essential in obese individuals
-
Even 5–10% weight loss significantly improves lipid profile
4. Smoking Cessation
-
Smoking lowers HDL and accelerates atherosclerosis
-
Cessation improves HDL and vascular health
II. Management of Secondary Causes
Secondary dyslipidemia must be corrected before drug therapy.
| Cause | Management |
|---|---|
| Diabetes mellitus | Glycemic control |
| Hypothyroidism | Thyroxine replacement |
| Nephrotic syndrome | Treat renal disease |
| Alcoholism | Alcohol restriction |
| Drug-induced | Modify or stop drug |
III. Pharmacological Therapy
Drug therapy is indicated when:
-
Lifestyle measures fail
-
High cardiovascular risk is present
-
Genetic dyslipidemia exists
1. Statins (First-Line Drugs)
Examples: Atorvastatin, Rosuvastatin
Mechanism:
-
Inhibit HMG-CoA reductase
-
↓ Cholesterol synthesis
-
↑ LDL receptor expression → ↓ LDL
Effects:
-
↓ LDL (major effect)
-
Mild ↓ TG
-
Mild ↑ HDL
Most effective drugs for cardiovascular risk reduction
2. Fibrates
Examples: Fenofibrate, Gemfibrozil
Mechanism:
-
Activate PPAR-α
-
↑ Lipoprotein lipase activity
Effects:
-
↓ Triglycerides (major effect)
-
↑ HDL
Indication:
-
Severe hypertriglyceridemia
-
Prevention of pancreatitis
3. Niacin (Nicotinic Acid)
Effects:
-
↓ LDL
-
↓ Triglycerides
-
↑ HDL (best drug to raise HDL)
Limitations:
-
Flushing
-
Hepatotoxicity
-
Insulin resistance
4. Ezetimibe
Mechanism:
-
Inhibits intestinal cholesterol absorption
Use:
-
Combined with statins when LDL targets not achieved
5. Bile Acid Sequestrants
Examples: Cholestyramine
Mechanism:
-
Bind bile acids → increased cholesterol excretion
Effect:
-
↓ LDL
6. PCSK9 Inhibitors
-
Increase LDL receptor availability
-
Used in familial hypercholesterolemia
IV. Management According to Lipid Abnormality
| Condition | Preferred Treatment |
|---|---|
| High LDL | Statins |
| High TG | Fibrates ± lifestyle |
| Low HDL | Exercise, niacin |
| Mixed dyslipidemia | Statin + fibrate (carefully) |
| Type I hyperlipoproteinemia | Diet + fibrates |
V. Monitoring of Therapy
-
Lipid profile repeated:
-
6–12 weeks after starting treatment
-
Every 6 months once stable
-
-
Monitor for:
-
Liver enzymes (statins)
-
Muscle symptoms
-
-
Adjust dose based on response
MCQs
1. Plasma lipids are transported in blood mainly as:
A. Free fatty acids
B. Micelles
C. Lipoproteins
D. Phospholipid bilayers
2. Major lipid carried by LDL is:
A. Triglyceride
B. Phospholipid
C. Cholesterol
D. Free fatty acid
3. HDL is primarily involved in:
A. Cholesterol delivery to tissues
B. Triglyceride transport
C. Reverse cholesterol transport
D. Fat digestion
4. Hyperlipoproteinemia refers to:
A. Low lipid levels
B. High lipoprotein levels
C. Normal lipid metabolism
D. Fat malabsorption
5. Hypolipoproteinemia is characterized by:
A. Increased LDL
B. Decreased lipoproteins
C. Increased triglycerides
D. Increased cholesterol
6. Primary lipid disorders are caused by:
A. Diet
B. Drugs
C. Genetic defects
D. Liver disease
7. Secondary lipid disorders are most commonly due to:
A. Apo-B mutation
B. PCSK9 mutation
C. Diabetes mellitus
D. LDL receptor defect
8. Fredrickson classification is based on:
A. Clinical features
B. Genetic mutations
C. Lipoprotein pattern
D. Enzyme levels
9. Type I hyperlipoproteinemia shows increase in:
A. LDL
B. VLDL
C. Chylomicrons
D. HDL
10. Type IIa hyperlipoproteinemia is characterized by:
A. ↑ VLDL
B. ↑ LDL
C. ↑ Chylomicrons
D. ↑ HDL
11. Most common primary hyperlipoproteinemia is:
A. Type I
B. Type IIa
C. Type IIb
D. Type V
12. Type III hyperlipoproteinemia is also called:
A. Familial hypercholesterolemia
B. Dysbetalipoproteinemia
C. Hyperchylomicronemia
D. Hypoalphalipoproteinemia
13. Palmar xanthomas are characteristic of:
A. Type I
B. Type IIa
C. Type III
D. Type IV
14. Type IV hyperlipoproteinemia shows increase in:
A. LDL
B. HDL
C. VLDL
D. Chylomicrons
15. High risk of pancreatitis is seen in:
A. Type IIa
B. Type IIb
C. Type I and Type V
D. Type III
16. Abetalipoproteinemia is associated with absence of:
A. Apo-A1
B. Apo-B
C. Apo-CII
D. Apo-E
17. Tangier disease is characterized by:
A. High LDL
B. High HDL
C. Very low HDL
D. High triglycerides
18. LCAT deficiency mainly affects:
A. LDL formation
B. Cholesterol esterification
C. Triglyceride synthesis
D. Fat absorption
19. Atherogenic lipoprotein is:
A. HDL
B. LDL
C. Chylomicron
D. VLDL only
20. Protective lipoprotein is:
A. LDL
B. IDL
C. HDL
D. VLDL
21. First step in atherosclerosis is:
A. Foam cell formation
B. Plaque rupture
C. Endothelial dysfunction
D. Thrombosis
22. Oxidized LDL leads to formation of:
A. Smooth muscle cells
B. Fibrous cap
C. Foam cells
D. Platelets
23. Earliest lesion of atherosclerosis is:
A. Fibrous plaque
B. Fatty streak
C. Calcified plaque
D. Thrombus
24. Foam cells are:
A. Smooth muscle cells
B. Lipid-laden macrophages
C. Endothelial cells
D. Fibroblasts
25. HDL protects against atherosclerosis by:
A. Increasing LDL
B. Oxidizing cholesterol
C. Reverse cholesterol transport
D. Increasing triglycerides
26. Basic test for lipid disorder evaluation is:
A. Apolipoprotein assay
B. Lipoprotein electrophoresis
C. Fasting lipid profile
D. Genetic testing
27. Fasting required for lipid profile is:
A. 4–6 hours
B. 6–8 hours
C. 9–12 hours
D. 24 hours
28. LDL-cholesterol is calculated using:
A. Friedewald formula
B. Beer–Lambert law
C. Michaelis–Menten equation
D. Henderson–Hasselbalch equation
29. Friedewald formula is invalid when triglycerides are:
A. <150 mg/dL
B. >200 mg/dL
C. >400 mg/dL
D. <100 mg/dL
30. VLDL-cholesterol is calculated as:
A. TC − HDL
B. TG ÷ 2
C. TG ÷ 5
D. HDL ÷ 5
31. Best predictor of cardiovascular risk is:
A. Total cholesterol
B. LDL alone
C. Apo-B / Apo-A1 ratio
D. Triglycerides
32. Lipoprotein(a) is:
A. Diet dependent
B. Drug induced
C. Genetically determined
D. Vitamin dependent
33. Secondary hyperlipidemia is commonly seen in:
A. Hyperthyroidism
B. Diabetes mellitus
C. Anemia
D. Malnutrition
34. Hypothyroidism causes increase in:
A. HDL
B. LDL
C. Chylomicrons
D. Apo-A1
35. Primary aim of lipid disorder management is:
A. Cosmetic improvement
B. Weight gain
C. Cardiovascular risk reduction
D. Fat absorption
36. First-line management of dyslipidemia is:
A. Statins
B. Fibrates
C. Lifestyle modification
D. PCSK9 inhibitors
37. Statins act by inhibiting:
A. Lipoprotein lipase
B. HMG-CoA reductase
C. ACAT
D. CETP
38. Best drugs for lowering LDL are:
A. Fibrates
B. Niacin
C. Statins
D. Bile acids
39. Fibrates are mainly used to treat:
A. Hypercholesterolemia
B. Hypertriglyceridemia
C. Low HDL
D. Tangier disease
40. Niacin is best known for:
A. Lowering LDL only
B. Raising HDL
C. Lowering Lp(a) only
D. Increasing triglycerides
41. Ezetimibe reduces cholesterol by:
A. Increasing LDL receptors
B. Inhibiting intestinal absorption
C. Increasing bile acid excretion
D. Increasing HDL synthesis
42. PCSK9 inhibitors are used mainly in:
A. Type I hyperlipoproteinemia
B. Familial hypercholesterolemia
C. Tangier disease
D. Abetalipoproteinemia
43. Monitoring lipid therapy is usually done after:
A. 1 week
B. 2 weeks
C. 6–12 weeks
D. 1 year
44. High TG with low HDL is commonly seen in:
A. Hypothyroidism
B. Diabetes mellitus
C. Anemia
D. Liver failure
45. Secondary causes of dyslipidemia must be:
A. Ignored
B. Treated first
C. Treated later
D. Never evaluated
46. Atherogenic index is calculated using:
A. LDL only
B. HDL only
C. Lipid ratios
D. Triglycerides only
47. Low HDL is associated with:
A. Reduced CV risk
B. Increased CV risk
C. No risk
D. Improved prognosis
48. Major complication of severe hypertriglyceridemia is:
A. Stroke
B. Pancreatitis
C. Hypertension
D. Anemia
49. Dyslipidemia affects dental practice mainly by:
A. Increasing caries
B. Delaying wound healing
C. Causing tooth discoloration
D. Increasing saliva
50. Most important lipid abnormality in atherosclerosis is:
A. High HDL
B. Low triglycerides
C. High LDL
D. Low VLDL
Answer Key
-
C
-
C
-
C
-
B
-
B
-
C
-
C
-
C
-
C
-
B
-
C
-
B
-
C
-
C
-
C
-
B
-
C
-
B
-
B
-
C
-
C
-
C
-
B
-
B
-
C
-
C
-
C
-
A
-
C
-
C
-
C
-
C
-
B
-
B
-
C
-
C
-
B
-
C
-
B
-
B
-
B
-
B
-
C
-
B
-
B
-
C
-
B
-
B
-
B
-
C
