Introduction
- Haemophilus is a genus of small, pleomorphic, Gram-negative bacteria commonly found in the human upper respiratory tract.
-
Members of this genus may exist as normal commensals or act as pathogenic organisms under suitable conditions.
-
The most important pathogenic species is Haemophilus influenzae, which is responsible for a variety of respiratory and invasive infections.
-
Haemophilus species are fastidious organisms, requiring special growth factors for cultivation in the laboratory.
-
Infections caused by Haemophilus are more common in infants, young children, elderly individuals, and immunocompromised patients.
-
Capsulated strains, especially type b, are associated with severe systemic infections such as meningitis and septicemia.
-
Haemophilus infections spread mainly through respiratory droplets.
-
Due to its clinical importance, Haemophilus is a significant organism in medical microbiology, particularly for MLT and blood bank students.
-
Early diagnosis, appropriate antibiotic therapy, and effective vaccination are essential for prevention and control of Haemophilus infections.
General Character
- Family: Pasteurellaceae
- Gram reaction: Gram-negative
- Morphology:
- Small, pleomorphic bacilli
- Often appear as coccobacilli
- Arrangement:
- Single cells
- Pairs or small groups
- Motility: Non-motile
- Spore formation: Non-spore forming
- Oxygen requirement:
- Facultative anaerobes
- Growth nature:
- Fastidious organisms
- Require enriched media
- Special growth factors:
- X factor (Hemin)
- V factor (NAD / NADP)
- Capsule:
- Present in some strains (H. influenzae type b)
- Important virulence factor
- Biochemical nature:
- Oxidase positive
- Catalase positive
- Habitat:
- Normal flora of upper respiratory tract (some species)
Morphology
-
Gram reaction:
-
Gram-negative
-
Appear pink on Gram staining
-
-
Shape:
-
Small pleomorphic bacilli
-
Often seen as coccobacilli
-
-
Size:
-
Very small bacteria (tiny rods)
-
-
Arrangement:
-
Occur as single cells
-
May be seen in pairs or small groups
-
-
Capsule:
-
Present in some species (e.g., Haemophilus influenzae type b)
-
Capsule is an important virulence factor
-
-
Motility:
-
Non-motile
-
-
Spore formation:
-
Non-spore forming
-
-
Pleomorphism:
-
Show variation in shape and size, especially in clinical specimens
-
Cultural Characteristics
-
Growth requirement:
-
Fastidious organism (requires enriched media)
-
-
Essential growth factors:
-
X factor (Hemin) – required for heme synthesis
-
V factor (NAD / NADP) – required for growth and metabolism
-
-
Ordinary media:
-
Does not grow on simple media like nutrient agar
-
-
Chocolate agar:
-
Best medium for growth
-
Heating releases X and V factors
-
Produces small, smooth, moist, translucent colonies
-
-
Blood agar:
-
H. influenzae does not grow alone
-
Shows satellite phenomenon around Staphylococcus aureus
-
Staphylococcus provides V factor
-
-
-
Selective media:
-
Chocolate agar with antibiotics (e.g., bacitracin) used for throat specimens
-
-
Colony morphology:
-
Small, convex, grayish, dew-drop like colonies
-
Capsulated strains form larger, mucoid colonies
-
-
Atmosphere:
-
Grows best in 5–10% CO₂ (capnophilic)
-
-
Temperature:
-
Optimal growth at 35–37°C
-
Biochemical Reactions
General Biochemical Characteristics
-
Oxidase test: Positive
-
Catalase test: Positive (most species)
-
Indole test: Variable (species dependent)
-
Urease test: Negative
-
Nitrate reduction: Positive
Carbohydrate Fermentation
-
Fermentation occurs without gas production
| Species | Glucose | Sucrose | Lactose | Indole |
|---|---|---|---|---|
| H. influenzae | + | – | – | Variable |
| H. parainfluenzae | + | + | – | + |
| H. ducreyi | – | – | – | – |
| H. aegyptius | + | – | – | – |
Other Important Tests
-
Porphyrin test (ALA test):
-
H. influenzae → Negative (requires X factor)
-
H. parainfluenzae → Positive (does not require X factor)
-
-
ONPG test: Negative
Pathogenicity
Virulence Factors
-
Capsule
-
Present in capsulated strains (especially H. influenzae type b – Hib)
-
Prevents phagocytosis
-
Major virulence factor
-
-
Lipooligosaccharide (LOS)
-
Acts as endotoxin
-
Causes inflammation and tissue damage
-
-
IgA protease
-
Destroys secretory IgA
-
Helps in colonization of respiratory mucosa
-
-
Fimbriae (Pili)
-
Help in adherence to epithelial cells
-
-
Outer membrane proteins (OMP)
-
Aid in attachment and immune evasion
-
Pathogenesis
-
Entry through respiratory or genital mucosa
-
Adherence to epithelial cells using pili
-
Local multiplication
-
Invasion of bloodstream (mainly capsulated strains)
-
Spread to distant organs → meninges, lungs, joints
Diseases Caused
Haemophilus influenzae
-
Upper respiratory tract infections
-
Otitis media
-
Sinusitis
-
Epiglottitis
-
-
Lower respiratory tract infections
-
Pneumonia
-
-
Invasive diseases
-
Meningitis (Hib)
-
Septicemia
-
Septic arthritis
-
Haemophilus ducreyi
-
Chancroid
-
Painful genital ulcer
-
Soft, non-indurated edges
-
Painful inguinal lymphadenitis (bubo)
-
Haemophilus aegyptius
-
Acute conjunctivitis (pink eye)
Host Factors
-
More common in:
-
Children
-
Immunocompromised individuals
-
Unvaccinated persons
-
Laboratory Diagnosis
1. Specimen Collection
-
Haemophilus influenzae
-
Sputum
-
Throat swab
-
Blood
-
Cerebrospinal fluid (CSF)
-
-
Haemophilus ducreyi
-
Pus or exudate from genital ulcer
-
-
Haemophilus aegyptius
-
Conjunctival swab
-
2. Direct Microscopy
-
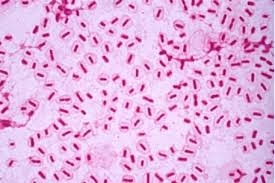
Gram staining
-
Small Gram-negative pleomorphic bacilli / coccobacilli
-
-
CSF examination
-
Shows Gram-negative bacilli in meningitis cases
-
3. Culture
-
Chocolate agar
-
Medium of choice
-
Incubation at 35–37°C, 5–10% CO₂
-
-
Blood agar
-
Shows satellite phenomenon around Staphylococcus aureus
-
-
Selective media
-
Chocolate agar with bacitracin (respiratory specimens)
-
4. Identification Tests
-
X and V factor requirement
-
H. influenzae → requires both X & V
-
-
Porphyrin (ALA) test
-
Negative in H. influenzae
-
-
Biochemical tests
-
Oxidase: Positive
-
Catalase: Positive
-
Sugar fermentation pattern for species identification
-
5. Serological Tests
-
Capsular antigen detection
-
Latex agglutination test (CSF)
-
-
Useful for Hib meningitis
6. Molecular Methods
-
PCR
-
Rapid and sensitive detection
-
Useful in culture-negative cases
-
7. Antimicrobial Sensitivity Testing
-
Performed due to:
-
β-lactamase producing strains
-
-
Helps in guiding therapy
Antibiotic Resistance
Mechanisms of Resistance
-
β-lactamase production
-
Most common mechanism
-
Causes resistance to ampicillin and penicillin
-
Enzyme breaks β-lactam ring
-
-
Altered Penicillin-Binding Proteins (PBPs)
-
Seen in β-lactamase–negative ampicillin-resistant (BLNAR) strains
-
Reduced affinity for β-lactam antibiotics
-
-
Efflux pumps
-
Contribute to resistance against macrolides and tetracyclines
-
-
Target site modification
-
Causes resistance to fluoroquinolones (rare but reported)
-
Commonly Resistant Antibiotics
-
Ampicillin
-
Penicillin
-
First-generation cephalosporins
-
Macrolides (in some strains)
Usually Effective Antibiotics
-
Amoxicillin + clavulanic acid
-
Second and third-generation cephalosporins
-
Cefuroxime
-
Ceftriaxone
-
-
Fluoroquinolones (adults)
-
Azithromycin (variable sensitivity)
Detection of Resistance
-
β-lactamase test
-
Nitrocefin test (rapid)
-
-
Antibiotic sensitivity testing
-
Disk diffusion / MIC methods
-
Prevention
1. Vaccination
-
Hib vaccine (Haemophilus influenzae type b)
-
Most effective preventive measure
-
Prevents meningitis, pneumonia, epiglottitis, septicemia
-
Given in infancy as part of routine immunization
-
Available as pentavalent / hexavalent vaccines
-
2. Chemoprophylaxis
-
Rifampicin
-
Given to close contacts of Hib meningitis cases
-
Prevents secondary spread
-
Indicated for:
-
Household contacts
-
Daycare contacts
-
-
3. Infection Control Measures
-
Proper hand hygiene
-
Respiratory etiquette (covering cough/sneeze)
-
Isolation of infected patients when required
4. Early Diagnosis and Treatment
-
Prompt identification and appropriate antibiotic therapy
-
Reduces transmission and complications
5. Public Health Measures
-
Routine childhood immunization programs
-
Surveillance of antibiotic resistance
-
Health education and awareness
6. Prevention of Sexually Transmitted Infection (H. ducreyi)
-
Safe sexual practices
-
Early diagnosis and treatment of chancroid
-
Partner notification and treatment
MCQs
1. Haemophilus belongs to which family?
A. Enterobacteriaceae
B. Neisseriaceae
C. Pasteurellaceae
D. Vibrionaceae
2. Haemophilus influenzae is a:
A. Gram-positive coccus
B. Gram-negative bacillus
C. Gram-positive bacillus
D. Acid-fast bacillus
3. Morphology of Haemophilus is best described as:
A. Cocci in chains
B. Long rods
C. Pleomorphic coccobacilli
D. Spiral shaped
4. Haemophilus species are:
A. Obligate anaerobes
B. Obligate aerobes
C. Facultative anaerobes
D. Microaerophilic
5. Which factor is required by H. influenzae for growth?
A. Only X factor
B. Only V factor
C. Both X and V factors
D. Neither X nor V factor
6. X factor is chemically:
A. NAD
B. Hemin
C. Riboflavin
D. Biotin
7. V factor is:
A. Hemin
B. Iron
C. NAD / NADP
D. Vitamin K
8. Best culture medium for Haemophilus is:
A. Nutrient agar
B. MacConkey agar
C. Chocolate agar
D. TCBS agar
9. Satellite phenomenon is seen on:
A. Chocolate agar
B. Blood agar
C. MacConkey agar
D. Lowenstein-Jensen medium
10. Satellite phenomenon occurs due to:
A. X factor release
B. V factor release by Staphylococcus
C. Capsule production
D. Hemolysis
11. Haemophilus influenzae is oxidase:
A. Negative
B. Positive
C. Variable
D. Not done
12. Catalase reaction of Haemophilus is usually:
A. Negative
B. Weak positive
C. Positive
D. Variable
13. Which sugar is fermented by H. influenzae?
A. Lactose
B. Sucrose
C. Glucose
D. Mannitol
14. Gas production from sugars by Haemophilus is:
A. Present
B. Absent
C. Variable
D. Depends on media
15. Porphyrin (ALA) test in H. influenzae is:
A. Positive
B. Negative
C. Weakly positive
D. Variable
16. Capsule of H. influenzae is made of:
A. Protein
B. Lipid
C. Polysaccharide
D. Lipopolysaccharide
17. Most virulent strain of H. influenzae is:
A. Non-typeable
B. Type a
C. Type b
D. Type f
18. Major virulence factor of H. influenzae is:
A. Flagella
B. Capsule
C. Exotoxin
D. Enzyme
19. Hib commonly causes meningitis in:
A. Elderly
B. Neonates
C. Children
D. Pregnant women
20. Haemophilus ducreyi causes:
A. Syphilis
B. Gonorrhea
C. Chancroid
D. LGV
21. Chancroid ulcer is characteristically:
A. Painless and hard
B. Painful and soft
C. Painless and soft
D. Hard with induration
22. Haemophilus aegyptius is associated with:
A. Pneumonia
B. Meningitis
C. Conjunctivitis
D. Otitis media
23. Habitat of Haemophilus influenzae is:
A. Skin
B. Intestine
C. Upper respiratory tract
D. Genital tract
24. Gram stain of Haemophilus shows:
A. Purple cocci
B. Pink bacilli
C. Blue spiral forms
D. Red acid-fast rods
25. Motility of Haemophilus is:
A. Motile
B. Non-motile
C. Peritrichous
D. Polar
26. Spore formation in Haemophilus:
A. Present
B. Absent
C. Seen in old cultures
D. Variable
27. Endotoxin of Haemophilus is:
A. LPS
B. LOS
C. Exotoxin A
D. Protein toxin
28. IgA protease helps in:
A. Capsule formation
B. Antibiotic resistance
C. Mucosal colonization
D. Spore formation
29. Drug of choice for Hib meningitis is:
A. Ampicillin
B. Penicillin
C. Ceftriaxone
D. Erythromycin
30. Most common resistance mechanism in Haemophilus is:
A. Efflux pump
B. Capsule change
C. β-lactamase production
D. Spore formation
31. BLNAR strains are:
A. β-lactamase positive
B. Ampicillin sensitive
C. β-lactamase negative but resistant
D. Fully sensitive
32. Rapid test to detect β-lactamase is:
A. Coagulase test
B. Oxidase test
C. Nitrocefin test
D. Indole test
33. Chemoprophylaxis drug for Hib contacts is:
A. Ciprofloxacin
B. Rifampicin
C. Penicillin
D. Doxycycline
34. Hib vaccine is composed of:
A. Live bacteria
B. Killed bacteria
C. Capsular polysaccharide
D. Protein toxoid
35. Hib vaccine is mainly protective against:
A. All strains
B. Type b strains
C. Non-typeable strains
D. H. ducreyi
36. Haemophilus grows best in presence of:
A. Oxygen only
B. CO₂ (5–10%)
C. Nitrogen
D. Anaerobic jar
37. Colony morphology on chocolate agar is:
A. Large dry colonies
B. Small moist translucent colonies
C. Swarming growth
D. Pigmented colonies
38. Haemophilus does NOT grow on:
A. Chocolate agar
B. Blood agar alone
C. Enriched media
D. CO₂ incubator
39. Latex agglutination test detects:
A. Antibody
B. Capsule antigen
C. Endotoxin
D. Enzyme
40. Best specimen for suspected Hib meningitis is:
A. Urine
B. Stool
C. CSF
D. Throat swab
41. Haemophilus is normal flora of:
A. Intestine
B. Skin
C. Upper respiratory tract
D. Blood
42. ONPG test in Haemophilus is usually:
A. Positive
B. Negative
C. Variable
D. Weak positive
43. Haemophilus parainfluenzae requires:
A. X factor only
B. V factor only
C. Both X and V
D. Neither X nor V
44. Gas gangrene is caused by Haemophilus:
A. True
B. False
45. Haemophilus is fastidious because:
A. Slow growing
B. Needs enriched media
C. Produces spores
D. Anaerobic
46. Which test differentiates X-factor dependency?
A. Oxidase test
B. Indole test
C. Porphyrin test
D. Catalase test
47. Hib meningitis is common in:
A. Adults only
B. Children <5 years
C. Neonates only
D. Elderly only
48. Painful inguinal bubo is seen in:
A. Syphilis
B. Gonorrhea
C. Chancroid
D. LGV
49. Haemophilus is best described as:
A. Acid-fast bacillus
B. Gram-negative fastidious bacillus
C. Gram-positive cocci
D. Spore-forming rod
50. Most effective prevention against Hib is:
A. Antibiotics
B. Hygiene
C. Vaccination
D. Isolation
Answer Key
-
C
-
B
-
C
-
C
-
C
-
B
-
C
-
C
-
B
-
B
-
B
-
C
-
C
-
B
-
B
-
C
-
C
-
B
-
C
-
C
-
B
-
C
-
C
-
B
-
B
-
B
-
B
-
C
-
C
-
C
-
C
-
C
-
B
-
C
-
B
-
B
-
B
-
B
-
B
-
C
-
C
-
B
-
B
-
B
-
B
-
C
-
B
-
C
-
B
-
C