Introduction
-
Enterobacter cloacae is a Gram-negative, rod-shaped bacterium belonging to the family Enterobacteriaceae
-
It is widely distributed in nature and is commonly present as part of the normal intestinal flora of humans
-
The organism is a facultative anaerobe, capable of growth in both aerobic and anaerobic conditions
-
Enterobacter cloacae acts mainly as an opportunistic pathogen
-
It is an important cause of hospital-acquired (nosocomial) infections
-
Infections are more common in immunocompromised patients, neonates, and those with prolonged hospital stay
-
The organism is frequently associated with urinary tract infections, pneumonia, septicemia, wound infections, and intra-abdominal infections
-
Its ability to acquire multidrug resistance, especially through AmpC β-lactamase production, makes treatment challenging
General Character
Genus
Enterobacter
Species
Enterobacter cloacae
Family
Enterobacteriaceae
Gram staining
Enterobacter species are Gram-negative bacteria and appear pink on Gram staining due to the presence of a thin peptidoglycan layer and an outer membrane.
Shape and arrangement
-
Shape: Rod-shaped (bacilli)
-
Arrangement: Usually present as single cells, but may also occur in pairs or short chains
Oxygen requirements
Enterobacter species are facultative anaerobes, capable of growth under both aerobic and anaerobic conditions.
Morphology
Enterobacter cloacae is a Gram-negative bacillus with characteristic morphological features that aid in its identification in the microbiology laboratory.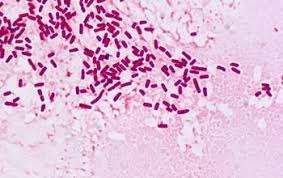
Shape and size
-
Rod-shaped (bacilli)
-
Medium-sized organisms measuring approximately 1–3 µm in length and 0.5–0.8 µm in width
Gram staining
-
Gram-negative
-
Appears pink on Gram staining due to a thin peptidoglycan layer and an outer membrane
Arrangement
-
Commonly seen as single cells
-
May also occur in pairs or short chains
Motility
-
Motile organism
-
Possesses peritrichous flagella
Capsule
-
Usually non-capsulated, though some strains may produce a thin capsule
Spores
-
Non-spore forming
Special morphological features
-
Presence of fimbriae (pili) which aid in adhesion
-
Typical cell wall structure of Enterobacteriaceae containing lipopolysaccharide (LPS)
Cultural Characteristics
Enterobacter cloacae grows readily on routine laboratory media and shows characteristic colony morphology helpful for identification.
Growth requirements
-
Facultative anaerobe
-
Grows well under aerobic and anaerobic conditions
-
Optimum temperature for growth is 37°C
-
Does not require special growth factors
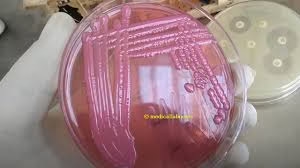
Nutrient agar
-
Produces large, smooth, moist, and greyish-white colonies
-
Colonies are convex with regular margins
-
Surface appears glistening
Blood agar
-
Forms large, smooth, moist colonies
-
Usually non-hemolytic (gamma hemolysis)
MacConkey agar
-
Lactose fermenter
-
Colonies appear pink, often mucoid due to capsule formation
-
May show late or variable lactose fermentation in some strains
EMB agar
-
Produces pink to purple colonies
-
Does not show metallic sheen (distinguishes from E. coli)
CLED agar
-
Shows yellow colonies due to lactose fermentation
-
Commonly isolated from urinary tract infections
Odor
-
No characteristic or distinctive odor
Biochemical Reactions
The biochemical reaction pattern of Enterobacter cloacae helps in its identification and differentiation from other members of the family Enterobacteriaceae.
Oxidase test
-
Negative
Catalase test
-
Positive
Indole test
-
Negative
Methyl red test
-
Negative
Voges–Proskauer test
-
Positive
Citrate utilization test
-
Positive
Urease test
-
Negative (occasionally weak positive)
Carbohydrate fermentation
-
Ferments glucose with acid and gas production
-
Lactose fermenter (often late fermenter)
-
Ferments mannitol and sucrose
Triple sugar iron (TSI) agar
-
Acid slant / acid butt (A/A)
-
Gas production present
-
No H₂S production
Nitrate reduction test
-
Positive (reduces nitrate to nitrite)
Motility test
-
Positive
ONPG test
-
Positive (confirms lactose fermentation)
Decarboxylation tests
-
Lysine decarboxylase: Negative
-
Ornithine decarboxylase: Positive
Pathogenicity
Enterobacter cloacae is an opportunistic pathogen that commonly causes hospital-acquired infections, particularly in patients with underlying illnesses or compromised immunity.
General pathogenic features
-
Part of the normal intestinal flora of humans
-
Causes disease mainly when host defenses are impaired
-
Frequently associated with nosocomial infections
Virulence factors
-
Lipopolysaccharide (LPS)
-
Acts as an endotoxin
-
Triggers fever, inflammation, and septic shock
-
-
Capsule (in some strains)
-
Inhibits phagocytosis
-
Enhances survival in host tissues
-
-
Fimbriae (pili)
-
Facilitate adhesion to epithelial cells and medical devices
-
-
Biofilm formation
-
Promotes persistence on catheters and hospital equipment
-
Contributes to chronic infections and antibiotic resistance
-
Clinical manifestations
Urinary tract infections
-
Commonly associated with catheterization
Respiratory tract infections
-
Pneumonia, especially in ICU and ventilated patients
Bloodstream infections
-
Bacteremia and septicemia, particularly in hospitalized or immunocompromised patients
Wound and soft tissue infections
-
Post-surgical and traumatic wound infections
Intra-abdominal infections
-
Peritonitis and abscesses, often following abdominal surgery
Neonatal infections
-
Sepsis and meningitis in neonates (rare but serious)
Laboratory Diagnosis
Specimen collection
-
Urine (urinary tract infections)
-
Blood (septicemia, bacteremia)
-
Sputum or endotracheal aspirate (pneumonia)
-
Pus or wound swab (surgical site and soft tissue infections)
-
Body fluids such as peritoneal fluid (intra-abdominal infections)
Direct microscopy
-
Gram staining shows Gram-negative bacilli
-
Presence of pus cells in infected specimens
Culture methods
Primary culture media
-
Nutrient agar: large, smooth, moist, greyish colonies
-
Blood agar: smooth, non-hemolytic colonies
-
MacConkey agar: lactose-fermenting pink colonies, often mucoid
Differential and confirmatory tests
-
EMB agar: pink to purple colonies without metallic sheen
-
CLED agar: yellow colonies in urine specimens
Biochemical identification
-
Oxidase: negative
-
Catalase: positive
-
Indole: negative
-
Methyl red: negative
-
Voges–Proskauer: positive
-
Citrate: positive
-
TSI: A/A with gas, no H₂S
-
Urease: negative
-
Motility: positive
-
ONPG: positive
Automated identification systems
-
VITEK, Phoenix, or MALDI-TOF MS for rapid species identification
Antibiotic susceptibility testing
-
Mandatory due to frequent multidrug resistance
-
Performed by Kirby–Bauer disc diffusion, MIC methods, or automated systems
Molecular methods (advanced laboratories)
-
PCR for detection of resistance genes during outbreaks
Antibiotic Resistance
Enterobacter cloacae is well known for its intrinsic and acquired antibiotic resistance, making infections difficult to treat, especially in hospital settings.
Intrinsic resistance
-
Naturally resistant to ampicillin and first-generation cephalosporins
-
Low outer membrane permeability reduces antibiotic entry
β-lactamase production
-
Produces chromosomal AmpC β-lactamase
-
AmpC confers resistance to:
-
Penicillins
-
First- and second-generation cephalosporins
-
-
AmpC may be inducible or derepressed, leading to treatment failure
Extended-spectrum β-lactamases (ESBLs)
-
Some strains acquire ESBLs, causing resistance to:
-
Third-generation cephalosporins
-
Aztreonam
-
Carbapenem resistance
-
Resistance due to:
-
Production of carbapenemases (e.g., KPC, NDM, OXA-48)
-
Reduced porin expression combined with AmpC or ESBL production
-
Non–β-lactam resistance mechanisms
-
Aminoglycoside-modifying enzymes
-
Target site mutations causing fluoroquinolone resistance
-
Overexpression of efflux pumps
Biofilm-associated resistance
-
Biofilm formation on catheters and devices reduces antibiotic penetration
-
Contributes to chronic and recurrent infections
Multidrug-resistant (MDR) strains
-
Increasingly reported in ICU and hospitalized patients
-
Limit therapeutic options and increase morbidity and mortality
Clinical significance
-
Empirical therapy often unreliable
-
Antibiotic susceptibility testing is essential before treatment
-
Requires strict antibiotic stewardship programs
Prevention
Prevention of Enterobacter cloacae infections mainly focuses on hospital infection control practices, proper patient care, and judicious use of antibiotics, as these infections are commonly nosocomial.
General preventive measures
-
Strict adherence to hand hygiene by healthcare workers
-
Proper aseptic techniques during invasive procedures
-
Regular cleaning and disinfection of hospital surfaces and equipment
Hospital infection control
-
Surveillance and early detection of hospital-acquired infections
-
Proper sterilization of surgical instruments and medical devices
-
Regular monitoring and maintenance of ventilators, catheters, and IV lines
Patient-related measures
-
Minimizing the duration of indwelling devices such as urinary catheters and intravenous lines
-
Proper care of surgical wounds and burn injuries
-
Isolation or cohorting of patients infected with multidrug-resistant strains
Antibiotic stewardship
-
Rational and restricted use of antibiotics
-
Avoidance of unnecessary broad-spectrum antibiotics
-
Periodic review of hospital antibiotic policies
Environmental control
-
Safe disposal of biomedical waste
-
Prevention of contamination of fluids, solutions, and hospital equipment
Community and vaccine status
-
No licensed vaccine available against Enterobacter cloacae
-
Emphasis on hygiene and infection control rather than immunization
MCQs
1. Enterobacter cloacae belongs to the family
A. Pseudomonadaceae
B. Vibrionaceae
C. Enterobacteriaceae
D. Neisseriaceae
Answer: C
2. Gram staining of Enterobacter cloacae shows
A. Gram-positive cocci
B. Gram-positive bacilli
C. Gram-negative bacilli
D. Gram-negative cocci
Answer: C
3. Shape of Enterobacter cloacae is
A. Coccus
B. Spiral
C. Rod
D. Filamentous
Answer: C
4. Oxygen requirement of Enterobacter cloacae is
A. Obligate aerobe
B. Obligate anaerobe
C. Facultative anaerobe
D. Microaerophilic
Answer: C
5. Arrangement of Enterobacter cloacae is commonly
A. Chains
B. Clusters
C. Single cells or short chains
D. Diplococci
Answer: C
6. Motility of Enterobacter cloacae is due to
A. Polar flagella
B. Peritrichous flagella
C. Pili
D. Capsule
Answer: B
7. Capsule in Enterobacter cloacae is
A. Always present
B. Always absent
C. Present in some strains
D. Thick and prominent
Answer: C
8. Spores in Enterobacter cloacae are
A. Present
B. Absent
C. Seen occasionally
D. Heat resistant
Answer: B
9. Growth temperature optimum for Enterobacter cloacae is
A. 25°C
B. 30°C
C. 37°C
D. 42°C
Answer: C
10. On nutrient agar, colonies are typically
A. Dry and rough
B. Smooth, moist, greyish
C. Pigmented
D. Swarming
Answer: B
11. Hemolysis on blood agar is usually
A. Alpha
B. Beta
C. Gamma (non-hemolytic)
D. Double zone
Answer: C
12. On MacConkey agar, Enterobacter cloacae forms
A. Colorless colonies
B. Pink lactose-fermenting colonies
C. Black colonies
D. Green colonies
Answer: B
13. Lactose fermentation in Enterobacter cloacae is
A. Rapid
B. Absent
C. Late or slow
D. Variable only in urine
Answer: C
14. EMB agar shows colonies that are
A. Colorless
B. Metallic sheen
C. Pink to purple without sheen
D. Black centered
Answer: C
15. Odor produced by Enterobacter cloacae is
A. Fruity
B. Grape-like
C. Putrid
D. No characteristic odor
Answer: D
16. Oxidase test of Enterobacter cloacae is
A. Positive
B. Negative
C. Weakly positive
D. Variable
Answer: B
17. Catalase test is
A. Negative
B. Positive
C. Variable
D. Weak
Answer: B
18. Indole test is
A. Positive
B. Negative
C. Variable
D. Weakly positive
Answer: B
19. Methyl red test is
A. Positive
B. Negative
C. Variable
D. Weak
Answer: B
20. Voges–Proskauer test is
A. Negative
B. Positive
C. Variable
D. Weak
Answer: B
21. Citrate utilization test is
A. Negative
B. Positive
C. Variable
D. Weak
Answer: B
22. Urease test is usually
A. Positive
B. Strongly positive
C. Negative
D. Rapidly positive
Answer: C
23. Glucose fermentation results in
A. Acid only
B. Acid and gas
C. Gas only
D. No fermentation
Answer: B
24. TSI reaction is typically
A. K/A with gas
B. A/A with gas
C. K/K
D. A/A with H₂S
Answer: B
25. Nitrate reduction test is
A. Negative
B. Positive
C. Variable
D. Weak
Answer: B
26. ONPG test is
A. Negative
B. Positive
C. Variable
D. Not done
Answer: B
27. Lysine decarboxylase is
A. Positive
B. Negative
C. Variable
D. Strongly positive
Answer: B
28. Ornithine decarboxylase is
A. Negative
B. Positive
C. Variable
D. Weak
Answer: B
29. Enterobacter cloacae is mainly a
A. Primary pathogen
B. Zoonotic pathogen
C. Opportunistic pathogen
D. Environmental contaminant only
Answer: C
30. Normal habitat of Enterobacter cloacae is
A. Skin
B. Upper respiratory tract
C. Intestinal tract
D. Blood
Answer: C
31. Most common infections caused are
A. Skin infections
B. UTIs and pneumonia
C. Gastroenteritis only
D. CNS infections only
Answer: B
32. Enterobacter cloacae infections are commonly
A. Community acquired
B. Food borne
C. Nosocomial
D. Vector borne
Answer: C
33. Major virulence factor is
A. Exotoxin
B. Capsule and endotoxin
C. Neurotoxin
D. Enterotoxin
Answer: B
34. Endotoxin present is
A. Teichoic acid
B. Capsule polysaccharide
C. Lipopolysaccharide
D. Protein A
Answer: C
35. Biofilm formation helps in
A. Sporulation
B. Motility
C. Antibiotic resistance
D. Pigment production
Answer: C
36. Most important resistance mechanism is
A. ESBL only
B. AmpC β-lactamase
C. Efflux pump only
D. Capsule formation
Answer: B
37. Enterobacter cloacae is intrinsically resistant to
A. Vancomycin
B. Ampicillin
C. Carbapenems
D. Colistin
Answer: B
38. Carbapenem resistance occurs due to
A. Capsule loss
B. Carbapenemase production
C. Flagellar mutation
D. Spore formation
Answer: B
39. MDR strains are common in
A. OPD
B. Community
C. ICU
D. Schools
Answer: C
40. Most important step before treatment is
A. Gram stain only
B. Culture only
C. Antibiotic susceptibility testing
D. Serology
Answer: C
41. Common specimen for diagnosis is
A. Throat swab
B. Urine
C. CSF only
D. Stool only
Answer: B
42. Gram stain from specimen shows
A. Gram-positive cocci
B. Gram-negative bacilli
C. Acid-fast bacilli
D. Yeast cells
Answer: B
43. Automated identification can be done by
A. ELISA
B. VITEK / MALDI-TOF
C. Latex agglutination
D. Weil–Felix test
Answer: B
44. Prevention mainly involves
A. Vaccination
B. Hand hygiene and asepsis
C. Chemoprophylaxis
D. Isolation only
Answer: B
45. Vaccine against Enterobacter cloacae is
A. Available
B. Under trial
C. Not available
D. Given to ICU patients
Answer: C
46. Catheter-associated UTIs are commonly due to
A. E. coli only
B. Proteus mirabilis
C. Enterobacter cloacae
D. Salmonella
Answer: C
47. Neonatal infections caused by Enterobacter cloacae include
A. Diarrhea only
B. Sepsis and meningitis
C. Skin rash
D. Otitis media
Answer: B
48. Enterobacter cloacae is oxidase
A. Positive
B. Negative
C. Variable
D. Weakly positive
Answer: B
49. Gas production is seen in
A. Lactose fermentation only
B. Glucose fermentation
C. Protein metabolism
D. Nitrate reduction
Answer: B
50. Most effective preventive strategy is
A. Vaccination
B. Early discharge
C. Antibiotic stewardship and infection control
D. Vitamin supplementation
Answer: C