Introduction
-
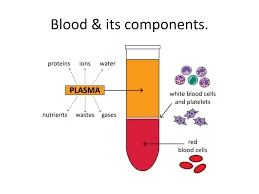
Whole blood contains:
-
Red Blood Cells (RBCs)
-
White Blood Cells (WBCs)
-
Platelets
-
Plasma (with clotting factors, proteins, electrolytes)
-
-
Modern transfusion practice follows the principle:
“Give only what the patient needs.”
-
Blood component preparation is performed in a licensed blood bank under sterile conditions.
-
It requires:
-
Blood collection bags with anticoagulant-preservative solution
-
Refrigerated centrifuge
-
Component extractor
-
Sealing device
-
Temperature-controlled storage units
-
Blood Collection
Types of Blood Donation
A. Whole Blood Donation
-
350 mL or 450 mL collected
-
Most common method
-
Used for component preparation
B. Apheresis Donation
-
Specific component collected (platelets, plasma, RBCs)
-
Remaining blood returned to donor
-
Requires automated apheresis machine
Donor Selection Criteria
Proper donor selection is mandatory to prevent transfusion-transmitted infections and protect donor health.
A. Age
-
18–65 years
B. Weight
-
Minimum 45 kg (for 350 mL)
-
≥ 55 kg (for 450 mL)
C. Hemoglobin
-
≥ 12.5 g/dL
D. Pulse
-
60–100/min (regular)
E. Blood Pressure
-
Systolic: 100–180 mmHg
-
Diastolic: 50–100 mmHg
F. Temperature
-
≤ 37.5°C
Donor Deferral
A. Temporary Deferral
-
Fever or infection
-
Recent vaccination
-
Recent surgery
-
Pregnancy
-
Recent blood donation (<3 months)
B. Permanent Deferral
-
HIV, HBV, HCV infection
-
Chronic renal disease
-
Malignancy
-
High-risk behavior
Pre-Donation Procedure
-
Registration
-
Medical history
-
Physical examination
-
Hemoglobin estimation
-
Informed consent
Blood Collection Equipment
-
Blood collection bag (single, double, triple, quadruple bag)
-
Sterile needle (16G)
-
Blood mixer with weighing scale
-
Anticoagulant-preservative solution (CPDA-1, CPD, SAGM)
-
Tourniquet
-
Sterile swabs
-
Sealing device
Blood Collection Bags
Types:
-
Single Bag
-
Used when whole blood is transfused directly
-
-
Double Bag
-
For the preparation of PRBC + Plasma
-
-
Triple Bag
-
For PRBC + Platelets + Plasma
-
-
Quadruple Bag
-
For PRBC + Platelets + Plasma + Cryoprecipitate
-
Anticoagulant-Preservative Solutions
A. CPD (Citrate Phosphate Dextrose)
-
Shelf life: 21 days
B. CPDA-1
-
Shelf life: 35 days
-
Contains adenine for ATP maintenance
C. SAGM (Additive solution)
-
Shelf life: 42 days
-
Improves RBC survival
Mechanism of Action:
-
Citrate → Binds calcium (prevents clotting)
-
Dextrose → Energy source
-
Adenine → Maintains ATP
-
Mannitol → Prevents hemolysis
-
Phosphate → Buffer
Procedure of Blood Collection
Step 1: Donor Position
-
Donor lies comfortably on donation couch
Step 2: Site Selection
-
Median cubital vein preferred
Step 3: Skin Preparation
-
Clean with:
-
Spirit
-
Povidone-iodine
-
Spirit again
-
-
Allow to dry
Step 4: Venepuncture
-
Use 16G sterile needle
-
Insert smoothly
Step 5: Collection
-
Blood flows into bag
-
Blood mixer ensures:
-
Continuous mixing
-
Correct volume
-
-
Collection time: 8–12 minutes
Step 6: Completion
-
Clamp tubing
-
Remove needle
-
Apply pressure dressing
Volume Collected
| Type | Volume Collected |
|---|---|
| 350 mL Bag | 350 ± 10% mL |
| 450 mL Bag | 450 ± 10% mL |
Anticoagulant ratio must be maintained properly.
Sample Collection for Testing
Small pilot tubes collected for:
-
ABO grouping
-
Rh typing
-
Crossmatching
-
Screening for:
-
HIV
-
HBsAg
-
HCV
-
VDRL
-
Malaria
-
Post-Donation Care
-
Donor rests for 10–15 minutes
-
Oral fluids given
-
Avoid heavy exercise for 24 hours
-
Remove bandage after 4–6 hours
Complications of Blood Donation
A. Local
-
Hematoma
-
Pain
B. Systemic
-
Vasovagal reaction
-
Dizziness
-
Hypotension
-
Rarely syncope
Transportation to Component Room
-
Blood labeled properly
-
Time of collection recorded
-
Transported at 20–24°C
-
Processed within 6–8 hours
Quality Control in Blood Collection
-
Correct volume
-
No clot formation
-
Proper labeling
-
Sterility maintained
-
Documentation completed
Principle of Component Separation
Blood component separation is based on the physical and biochemical differences between various elements of whole blood. The aim is to separate whole blood into its therapeutic components so that each patient receives only the required component.
1. Basic Concept
Whole blood contains:
- Red Blood Cells (RBCs)
- White Blood Cells (WBCs)
- Platelets
- Plasma
These components differ in:
- Density
- Size
- Weight
- Sedimentation rate
Separation is mainly achieved by centrifugation, which uses centrifugal force to separate components based on their density.
2. Principle of Centrifugation
Centrifugation works on the principle that:
When blood is rotated at high speed, heavier components move outward (bottom), and lighter components remain toward the top.
Order of settling (from bottom to top):
- Red Blood Cells (heaviest)
- Buffy coat (WBCs + platelets)
- Plasma (lightest)
3. Density of Blood Components
| Component | Approximate Density |
|---|---|
| RBCs | 1.09 g/mL |
| WBCs | 1.06–1.08 g/mL |
| Platelets | 1.03–1.04 g/mL |
| Plasma | 1.025 g/mL |
Because RBCs are densest, they settle at the bottom during centrifugation.
4. Types of Centrifugation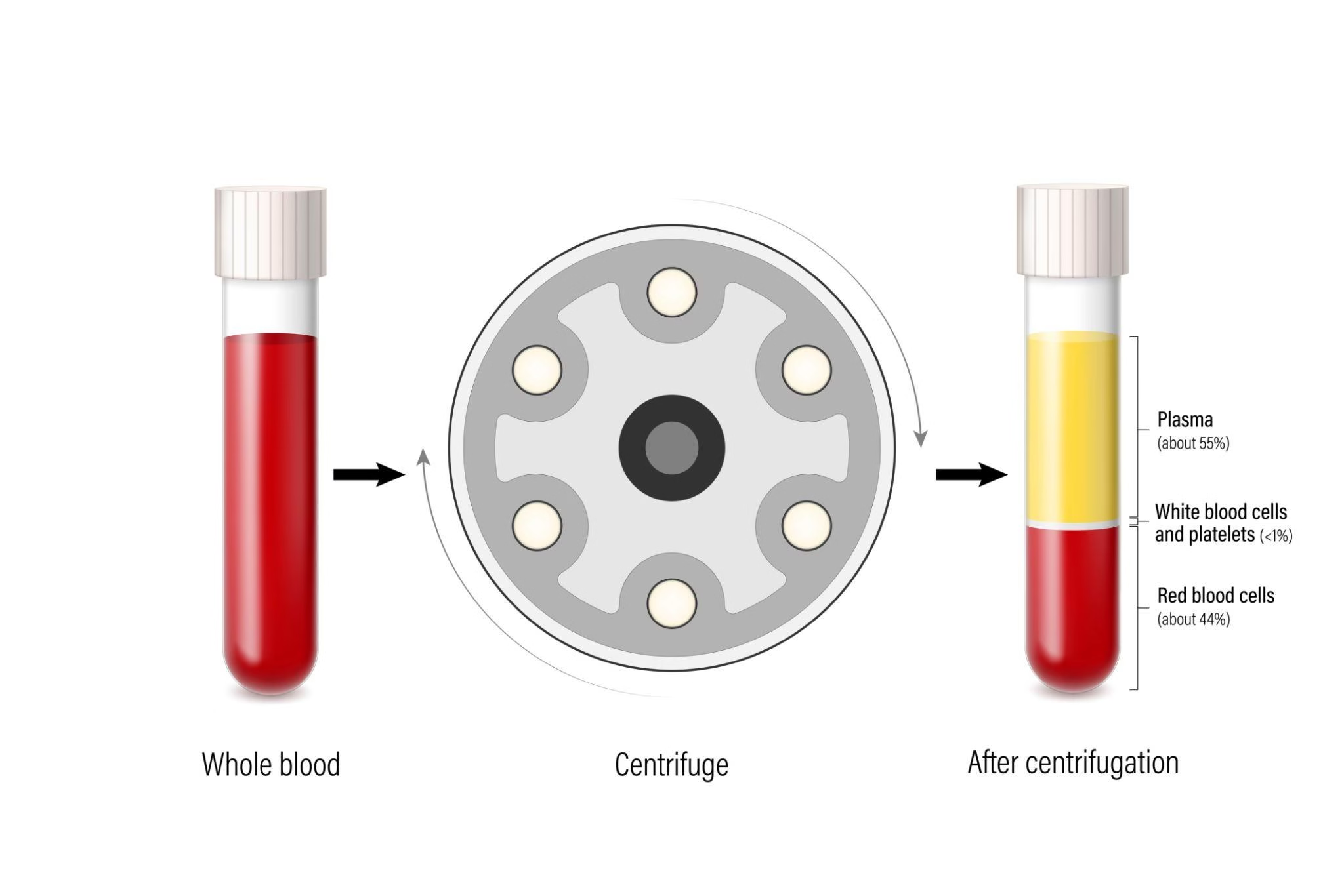
- Low speed centrifugation
- Separates:
- Packed RBCs (bottom)
- Platelet-Rich Plasma (PRP) (top)
Used in:
- Preparation of platelets
B. Hard Spin (Heavy Spin)
- High speed centrifugation
- Further separates PRP into:
- Platelet concentrate (bottom)
- Platelet-poor plasma (top)
Used in:
- Preparation of platelet concentrate and plasma products
5. Methods of Component Preparation
Two major methods are used:
A. Platelet-Rich Plasma (PRP) Method
- Soft spin → PRP separated
- Hard spin → Platelet concentrate formed
B. Buffy Coat Method
- Whole blood centrifuged at hard spin
- Buffy coat layer separated
- Platelets prepared from buffy coat
Buffy coat method gives:
- Higher platelet yield
- Better standardization
6. Principle of Plasma Separation
Plasma is the lightest component.
After centrifugation, it remains at the top.
It can be:
- Frozen immediately → Fresh Frozen Plasma (FFP)
- Further processed → Cryoprecipitate
7. Principle of Apheresis
Apheresis uses:
- Continuous flow centrifugation
- Selective removal of one component
- Remaining blood returned to donor
Principle:
- Automated density-based separation in real time
8. Important Factors Affecting Separation
- Centrifuge speed (RPM)
- Relative centrifugal force (RCF)
- Duration of centrifugation
- Temperature
- Type of anticoagulant
- Time between collection and processing
Improper centrifugation may cause:
- Hemolysis
- Poor platelet yield
- Reduced coagulation factor activity
9. Scientific Basis
Centrifugal force (F) depends on:
- Speed of rotation
- Radius of rotor
- Mass of particles
Heavier particles experience greater outward force and sediment faster.
10. Advantages of Component Separation
- Rational transfusion therapy
- Reduced volume overload
- Better inventory management
- Optimal utilization of one donation for multiple patients
Preparation of Packed Red Blood Cells
Procedure
Step 1: Collection of Whole Blood
-
350 mL or 450 mL collected
-
Anticoagulant used:
-
CPDA-1
-
CPD
-
SAGM (additive solution)
-
Step 2: First Centrifugation (Heavy Spin)
-
Whole blood placed in refrigerated centrifuge
-
Hard spin performed
Result:
-
RBCs settle at bottom
-
Plasma remains at top
-
Buffy coat layer in between
Step 3: Plasma Expression
-
Blood bag placed in plasma extractor
-
Plasma transferred into satellite bag
-
RBCs remain in primary bag
Step 4: Addition of Additive Solution (if used)
-
SAGM added to RBCs
-
Improves RBC survival
-
Extends shelf life
Characteristics of PRBCs
| Parameter | Value |
|---|---|
| Volume | 250–350 mL |
| Hematocrit | 55–65% |
| Hemoglobin content | ~50–70 g/unit |
| Plasma content | Minimal |
Storage Conditions
-
Temperature: 2–6°C
-
Storage refrigerator with continuous monitoring
Shelf Life:
| Anticoagulant | Shelf Life |
|---|---|
| CPD | 21 days |
| CPDA-1 | 35 days |
| SAGM | 42 days |
Biochemical Changes During Storage
During storage, RBCs undergo:
-
Decrease in ATP
-
Decrease in 2,3-DPG
-
Increased potassium leakage
-
Membrane rigidity
-
Hemolysis (minimal if proper storage)
These changes may affect oxygen delivery capacity.
Preparation of Platelet Concentrates
Types of Platelet Concentrates
-
Random Donor Platelets (RDP) – Prepared from whole blood
-
Single Donor Platelets (SDP) – Prepared by apheresis
PART A: Preparation of Random Donor Platelets (RDP)
-
Based on difference in density between RBCs, platelets, and plasma.
-
Prepared by two-step centrifugation:
-
Soft spin
-
Hard spin
-
Equipment Required
-
Refrigerated blood bank centrifuge
-
Triple or quadruple blood bag system
-
Plasma extractor
-
Platelet agitator (20–24°C)
-
Tube sealer
Procedure (PRP Method)
Step 1: Collection of Whole Blood
-
350 mL or 450 mL blood collected in CPDA-1/CPD bag
-
Blood processed within 6–8 hours
Step 2: First Centrifugation (Soft Spin)
-
Low-speed centrifugation
-
Purpose: Separate Platelet-Rich Plasma (PRP)
Result after soft spin:
-
Bottom: Packed RBCs
-
Top: Platelet-Rich Plasma (PRP)
Step 3: Transfer of PRP
-
Place bag on plasma extractor
-
Transfer PRP into satellite bag
-
RBCs remain in primary bag
Step 4: Second Centrifugation (Hard Spin)
-
High-speed centrifugation of PRP bag
Result:
-
Bottom: Platelet pellet
-
Top: Platelet-Poor Plasma (PPP)
Step 5: Expression of Plasma
-
Remove most of plasma
-
Leave about 40–70 mL plasma
-
Resuspend platelet pellet gently
Final product: Random Donor Platelet Concentrate
Final Characteristics of RDP
| Parameter | Value |
|---|---|
| Volume | 40–70 mL |
| Platelet count | ≥ 5.5 × 10¹⁰ per unit |
| WBC content | Variable |
| pH (end storage) | ≥ 6.2 |
Storage Conditions
-
Temperature: 20–24°C
-
Continuous gentle agitation
-
Shelf life: 5 days
Buffy Coat Method
-
Platelets concentrated from buffy coat layer
Steps
-
Whole blood centrifuged at hard spin
-
Buffy coat separated
-
Buffy coats pooled (4–6 donors)
-
Centrifuged again
-
Platelets suspended in plasma
Advantages:
-
Higher platelet yield
-
Better standardization
-
Reduced plasma volume
PART B: Preparation of Single Donor Platelets (SDP)
-
Continuous flow centrifugation
-
Selective removal of platelets
-
Remaining blood returned to donor
Equipment
-
Automated apheresis machine
-
Anticoagulant (ACD-A)
-
Sterile disposable tubing kit
Procedure
-
Donor selected and screened
-
Venous access established
-
Blood enters apheresis machine
-
Centrifugal separation occurs
-
Platelets collected
-
RBCs and plasma returned to donor
Final Characteristics of SDP
| Parameter | Value |
|---|---|
| Volume | 200–300 mL |
| Platelet count | ≥ 3.0 × 10¹¹ |
| Equivalent to | 5–6 RDP units |
| Donor exposure | Single donor |
Storage Conditions
-
20–24°C
-
Continuous agitation
-
Shelf life: 5 days
Quality Control Parameters
-
Platelet count adequate
-
pH ≥ 6.2
-
Swirling present (visual indicator of viability)
-
No clots
-
Sterility maintained
Preparation of Fresh Frozen Plasma
Procedure
Step 1: Collection of Whole Blood
-
350 mL or 450 mL blood collected
-
Anticoagulant: CPD or CPDA-1
-
Time of collection recorded carefully
Step 2: Centrifugation (Heavy Spin)
-
Whole blood centrifuged at appropriate speed
-
After centrifugation:
-
Bottom layer → Packed RBCs
-
Middle layer → Buffy coat
-
Top layer → Plasma
-
Step 3: Plasma Separation
-
Blood bag placed in plasma extractor
-
Plasma transferred into satellite bag
-
RBCs remain in primary bag
Care must be taken not to disturb the buffy coat.
Step 4: Rapid Freezing
-
Plasma must be frozen within 6–8 hours of collection
-
Temperature: –30°C or lower
Rapid freezing preserves:
-
Factor V
-
Factor VIII
-
Other coagulation factors
If frozen after 8 hours → It is called Plasma Frozen within 24 Hours (PF24), not FFP.
Step 5: Labeling
Label must include:
-
Component name (Fresh Frozen Plasma)
-
Blood group
-
Volume
-
Date of collection
-
Expiry date
-
Unique donor number
Characteristics of FFP
| Parameter | Value |
|---|---|
| Volume | 180–250 mL |
| Contains | All coagulation factors |
| Fibrinogen | 200–400 mg/unit |
| Plasma proteins | Albumin, globulin |
Storage Conditions
| Condition | Requirement |
|---|---|
| Temperature | –30°C or below |
| Shelf life | 1 year |
If stored at –18°C → Shelf life is 3 months (depending on guidelines).
Preparation of Cryoprecipitate
Cryoprecipitate is prepared from FFP.
Procedure:
Step 1: Selection of FFP
-
Use properly stored FFP
-
Ensure it was frozen within 6–8 hours of collection
-
Maintain temperature at –30°C before use
Step 2: Controlled Thawing
-
Thaw FFP at 1–6°C
-
Slow thawing allows formation of cold-insoluble precipitate
-
Duration: Approximately 12–18 hours
Result:
-
White cloudy precipitate forms at bottom
Step 3: Centrifugation
-
Perform cold centrifugation
-
Precipitate settles at bottom
-
Supernatant (cryosupernatant plasma) remains above
Step 4: Separation
-
Using plasma extractor:
-
Remove most of supernatant plasma
-
Leave 10–15 mL plasma with precipitate
-
This forms Cryoprecipitate
Step 5: Refreezing
-
Immediately refreeze cryoprecipitate
-
Store at –30°C or below
Characteristics of Cryoprecipitate
| Parameter | Value |
|---|---|
| Volume | 10–20 mL |
| Fibrinogen | ≥ 150–250 mg/unit |
| Factor VIII | ≥ 80 IU |
| Storage temperature | –30°C |
| Shelf life | 1 year |
Storage Conditions
-
Temperature: –30°C or lower
-
Shelf life: 1 year
-
Must not be thawed and refrozen repeatedly
Thawing Before Transfusion
-
Thawed at 30–37°C
-
Should be transfused within 6 hours
-
After pooling → Use within 4 hours
Leukocyte-Reduced Blood Components
Method:
-
Filtration using leukoreduction filters.
-
Removes ≥ 99% leukocytes.
Benefits:
-
Prevents febrile non-hemolytic reactions
-
Reduces CMV transmission
-
Prevents HLA alloimmunization
Washed Red Cells
Method:
-
RBCs washed with normal saline.
-
Removes plasma proteins.
Indications:
-
IgA deficiency
-
Recurrent allergic reactions
Irradiated Blood Components
Purpose:
-
Prevent Transfusion-Associated Graft vs Host Disease (TA-GVHD)
Method:
-
Gamma irradiation (25 Gy)
Indications:
-
Immunocompromised patients
-
Intrauterine transfusion
-
Bone marrow transplant recipients
Storage Requirements
| Component | Temperature | Shelf Life |
|---|---|---|
| PRBC | 2–6°C | 35–42 days |
| Platelets | 20–24°C | 5 days |
| FFP | ≤ -30°C | 1 year |
| Cryoprecipitate | ≤ -30°C | 1 year |
Quality Control of Blood Components
Each component must meet standards:
-
PRBC: Hematocrit 55–65%
-
Platelets: Adequate count per unit
-
FFP: Adequate factor levels
-
Sterility testing
Quality control is essential to ensure:
-
Safety
-
Potency
-
Efficacy
Advantages of Component Therapy
-
Rational use of blood
-
Reduced circulatory overload
-
Decreased transfusion reactions
-
Efficient inventory management
Complications if Improperly Prepared
-
Hemolysis
-
Bacterial contamination
-
Reduced factor activity
-
Platelet dysfunction
-
Transfusion reactions
