Introduction
- Lipids are a diverse group of organic biomolecules that are insoluble in water but soluble in non-polar organic solvents such as ether and chloroform.
-
They are mainly composed of carbon, hydrogen, and oxygen, and may also contain phosphorus and nitrogen.
-
Most lipids are formed by the esterification of fatty acids with alcohols and contain long hydrocarbon chains.
-
Lipids are generally hydrophobic, but some are amphipathic, having both hydrophilic and hydrophobic regions.
-
They are the most concentrated source of energy in the body, providing about 9 kcal per gram.
-
Lipids play an important role in energy storage, insulation, and protection of vital organs.
-
They are major structural components of biological membranes and help maintain membrane integrity and fluidity.
-
Knowledge of lipid chemistry is essential because abnormalities in lipid metabolism are associated with disorders such as obesity and cardiovascular diseases.
Properties of Lipids
-
Insolubility in Water – Lipids are hydrophobic and do not dissolve in water due to their long non-polar hydrocarbon chains.
-
Solubility in Organic Solvents – They dissolve readily in non-polar solvents such as ether, chloroform, benzene, and acetone.
-
High Energy Value – Lipids provide approximately 9 kcal per gram, making them the most energy-dense macronutrient.
-
Greasy/Oily Nature – Most lipids feel greasy to touch and leave a translucent stain on paper.
-
Lower Density than Water – Lipids are lighter than water and tend to float on its surface.
-
Melting Point Variation – Saturated fats have higher melting points (solid at room temperature), while unsaturated fats have lower melting points (liquid at room temperature).
-
Hydrolysis Property – Lipids can undergo hydrolysis to form fatty acids and alcohols in the presence of acids, alkali, or enzymes (lipases).
-
Oxidation (Rancidity) – Unsaturated lipids are prone to oxidation, leading to rancidity and unpleasant odor.
Biological Importance of Lipids
-
Major Source of Energy – Lipids are the most concentrated form of energy, providing about 9 kcal per gram, and serve as long-term energy reserves in the body.
-
Energy Storage – Stored as triglycerides in adipose tissue, lipids act as an efficient energy reservoir during fasting or starvation.
-
Structural Component of Cell Membranes – Phospholipids, glycolipids, and cholesterol are essential components of biological membranes, maintaining membrane structure and fluidity.
-
Thermal Insulation – Subcutaneous fat helps maintain body temperature by reducing heat loss.
-
Protection of Vital Organs – Adipose tissue cushions and protects organs such as the kidneys and heart from mechanical injury.
-
Absorption of Fat-Soluble Vitamins – Lipids are necessary for the absorption and transport of vitamins A, D, E, and K.
-
Precursor of Hormones and Signaling Molecules – Cholesterol serves as a precursor for steroid hormones, bile acids, and vitamin D; certain lipids also function as signaling molecules (e.g., prostaglandins).
-
Supply of Essential Fatty Acids – Essential fatty acids (like linoleic and linolenic acid) are required for normal growth, skin integrity, and proper cellular function.
Classification of Lipids

Bloor’s (1943) Classification
Simple Lipids
They are esters of fatty acids with alcohol
They are divided into two groups –
-
Fats and Oils (True Fats / Neutral Fats)
-
Esters of fatty acids + glycerol.
-
Solid at room temperature = fats (mainly in animals).
-
Liquid at room temperature = oils (mainly in plants).
-
Function: energy storage, insulation, protection.
-
-
Waxes
-
Esters of fatty acids + long-chain alcohols (not glycerol).
-
Found on plant leaves, skin, and feathers.
-
Function: waterproofing and protection.
-
Compound lipids
Fatty Acid + Alcohol (Glycerol/Sphingosine) + additional groups
They are divided into 4 groups –
-
Phospholipids
-
Structure: Glycerol (or sphingosine) + 2 fatty acids + phosphate group + nitrogenous base.
-
Amphipathic (hydrophilic head + hydrophobic tails).
-
Types:
-
Glycerophospholipids (phosphoglycerides) – contain glycerol.
-
Examples: Lecithin (phosphatidylcholine), Cephalin (phosphatidylethanolamine).
-
-
Sphingophospholipids – contain sphingosine.
-
Example: Sphingomyelin (important in myelin sheath of nerves).
-
-
-
Functions:
-
Structural role: essential in cell membranes (lipid bilayer).
-
Cell signaling: involved in signal transduction and membrane fluidity.
-
2. Glycolipids
-
Structure: Sphingosine + fatty acid + carbohydrate group.
-
Found mainly in the cell membranes of the brain and nerve cells.
-
Types:
-
Cerebrosides – contain a single sugar (glucose or galactose).
-
Gangliosides – contain complex oligosaccharides.
-
-
Functions:
-
Cell recognition and cell–cell interaction.
-
Important in nerve impulse transmission.
-
3. Lipoproteins
-
Structure: Lipid + protein complex.
-
Not structural like phospholipids, but serve as transport forms of lipids in the blood.
-
Types (based on density):
-
Chylomicrons – carry dietary triglycerides from intestine.
-
VLDL (Very Low-Density Lipoprotein) – transport triglycerides from liver.
-
LDL (Low-Density Lipoprotein) – carries cholesterol to tissues (“bad cholesterol”).
-
HDL (High-Density Lipoprotein) – carries cholesterol away to liver for excretion (“good cholesterol”).
-
-
Functions:
-
Transport of lipids (triglycerides, cholesterol) through the bloodstream.
-
-
4. Other Complex Lipids
1. Sulfolipids
-
-
Structure: Lipids containing sulfur groups, usually a sulfonic acid group (-SO₃H) attached to a sugar.
-
Example: Sulfoquinovosyl diacylglycerol (found in plants, algae, and bacteria).
-
Function:
-
Important in photosynthetic membranes (chloroplasts).
-
Found in bacterial cell walls, contributing to cell survival and virulence.
-
-
2. Aminolipids
-
-
Structure: Lipids that contain amino groups (–NH₂) in their structure.
-
Common in bacterial membranes.
-
Function:
-
Maintain membrane stability.
-
Help bacteria adapt to environmental stress (e.g., low pH).
-
Involved in interactions with host cells in pathogenic bacteria.
-
-
3. Lipo-polysaccharides (LPS)
-
-
Structure: A complex of lipid + polysaccharide.
-
Found in the outer membrane of Gram-negative bacteria.
-
Composed of:
-
Lipid A (toxic part).
-
Core polysaccharide.
-
O-antigen (antigenic determinant).
-
-
Function:
-
Provide structural integrity to bacterial outer membrane.
-
Protect bacteria from hostile environments.
-
Act as endotoxins in humans → cause fever, shock, and immune responses.
-
-
Derived Lipids or Precursor Lipids
Derived lipids are substances obtained by hydrolysis of simple and complex lipids. They are insoluble in water but soluble in organic solvents.
Examples of Derived Lipids:
-
-
Fatty acids – saturated (palmitic, stearic) & unsaturated (oleic, linoleic).
-
Glycerol – backbone of fats & oils.
-
Steroids – cholesterol, bile salts, steroid hormones (testosterone, estrogen, cortisol).
-
Terpenes & Carotenoids – vitamins A, E, K; plant pigments.
-
Eicosanoids – prostaglandins, leukotrienes, thromboxanes (act as local hormones).
-
Alcohols – long-chain alcohols (e.g., sphingosine).
-
Fat-soluble vitamins – A, D, E, K (classified as isoprenoid derivatives).
-
Miscellaneous Lipids
These are lipids that do not fit neatly into the categories of simple, complex, or derived lipids. They are diverse in structure and function.
Examples:
-
-
Carotenoids – plant pigments (β-carotene, lycopene); precursors of vitamin A.
-
Squalene – a triterpene, precursor of cholesterol and steroid hormones.
-
Eicosanoids – prostaglandins, thromboxanes, leukotrienes (local hormones).
-
Polyisoprenoids – dolichol, ubiquinone (coenzyme Q); involved in electron transport and glycoprotein synthesis.
-
Fatty acids
- Fatty acids are organic molecules that are long-chained carboxylic acids with 4-36 carbon atoms.
- Naturally occurring fatty acids are mostly unbranched, occurring in three main classes of lipids: triglycerides, phospholipids, and cholesteryl esters.
- Fatty acids are not found in the free state but remain associated with alcohol to form triglycerides.
- Fatty acids are stored as an energy reserve (fat) through an ester linkage to glycerol to form triglycerides.
Classification of Fatty Acids
Fatty acids are classified into four major classes
- Straight-chain fatty acids
- Branched-chain fatty acids
- Substituted fatty acids
- Cyclic fatty acids
1 Straight Chain Fatty
Acids Fatty acids, in which the carbons are arranged linearly, are subclassified into two classes:
-
- Saturated fatty acids
- Unsaturated fatty acids
Saturated fatty acids
There is no double bond in the hydrocarbon chain of these fatty acids. Saturated fatty acids are subclassified into two classes:
-
-
- Even carbon acids carry an even number of carbons,
-
e.g. palmitic acid and stearic acid.
-
-
- Odd carbon acids carry an odd number of carbons,
-
e.g. propionic acid.
Unsaturated fatty acids
These contain double bonds in their hydrocarbon chains.
These are subclassified according to the number of double bonds present in the structure as follows:
-
- Monoenoic or monounsaturated fatty acid
- Polyenoic or polyunsaturated fatty acid.
- Monoenoic or monounsaturated fatty acids, e.g. oleic acid.
- Polyenoic or polyunsaturated fatty acids, for example:
-
-
- Dienoic acids
- Trienoic acids,
- Tetraenoic acid,
-
2. Branched Chain Fatty Acids
These are less abundant than straight-chain acids in animals and plants, e.g.
-
- Isovaleric acid
- Isobutyric acid.
3. Substituted Fatty Acids
In substituted fatty acids, one or more hydrogen atoms have been replaced by another group, e.g.
-
- Lactic acid of blood.
- Cerebronic acid and oxynervonic acids of brain glycolipids.
- Ricinoleic acid of castor oil.
4. Cyclic Fatty Acids
Fatty acids bearing cyclic groups are present in some bacteria and seed lipids.
Functions of Fatty Acids
Fatty acids have three major physiological functions.
- They serve as building blocks of phospholipids and glycolipids. These amphipathic molecules are important components of biological membranes.
- Fatty acid derivatives serve as hormones, e.g. prostaglandins.
- Fatty acids serve as a major fuel for most cells.
Essential Fatty Acid
-
Essential fatty acids are fatty acids that cannot be synthesized by the human body and must be obtained through the diet.
-
They are mainly polyunsaturated fatty acids containing two or more double bonds in their structure.
-
The two primary essential fatty acids are linoleic acid (omega-6) and alpha-linolenic acid (omega-3).
-
They are considered essential because humans lack the enzymes required to introduce double bonds beyond the ninth carbon atom.
-
Essential fatty acids are important for maintaining the structure and fluidity of cell membranes.
-
They act as precursors for biologically active compounds such as prostaglandins, thromboxanes, and leukotrienes.
-
Major dietary sources include vegetable oils, nuts, seeds, and green leafy vegetables.
-
Deficiency may cause dry scaly dermatitis, growth retardation, poor wound healing, and increased susceptibility to infections.
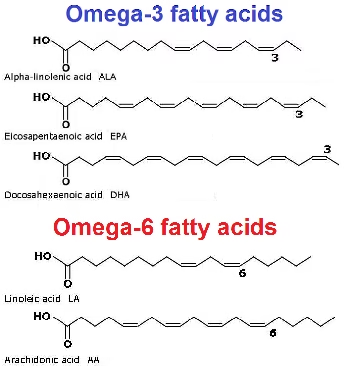
Functions of Essential Fatty Acids
-
Cell membrane structure – maintain fluidity and permeability.
-
Precursors of eicosanoids – prostaglandins, thromboxanes, leukotrienes (regulate inflammation, blood pressure, clotting).
-
Growth & development – important for brain and vision (especially in infants).
-
Lipid transport – help in formation of lipoproteins.
-
Skin health – prevent dryness and scaling.
-
Cholesterol metabolism – lower plasma cholesterol by promoting its excretion.
Deficiency of Essential Fatty Acids
-
Poor growth & development (especially in infants/children).
-
Skin problems – dryness, scaliness, dermatitis.
-
Reproductive failure.
-
Impaired wound healing.
-
Kidney & liver problems.
-
Neurological issues – learning difficulties, vision problems (due to brain/retina effects).
-
Increased risk of infections (weakened immunity).
Significance of ω3 Fatty Acid
- Decrease the risk of cardiovascular disease.
- Appear to replace arachidonic acid in platelet membranes.
- Lower the production of Thromboxane and tendency of the platelet aggregation.
- Decrease Serum Triglycerides.
- Important for Infant Development.
- Lower the risk of various mental illnesses (Depression, ADHD)
- Lower the risk of chronic degenerative diseases such as Cancer, Rheumatoid Arthritis, and Alzheimer’s Disease.
Triglycerides
-
Triglycerides, also known as triacylglycerols, are the main form of stored fat in the body.
-
They are formed by the esterification of one molecule of glycerol with three molecules of fatty acids.
-
Triglycerides are neutral lipids and are highly hydrophobic in nature.
-
They serve as the major energy reserve, providing approximately 9 kcal per gram.
-
They are stored primarily in adipose tissue and released during periods of fasting or increased energy demand.
-
The physical state of triglycerides depends on the type of fatty acids present; saturated fats are solid, while unsaturated fats are liquid at room temperature.
-
In the bloodstream, triglycerides are transported within lipoproteins such as chylomicrons and very low-density lipoproteins (VLDL).
-
Elevated triglyceride levels are associated with metabolic disorders such as obesity, diabetes mellitus, and cardiovascular diseases.
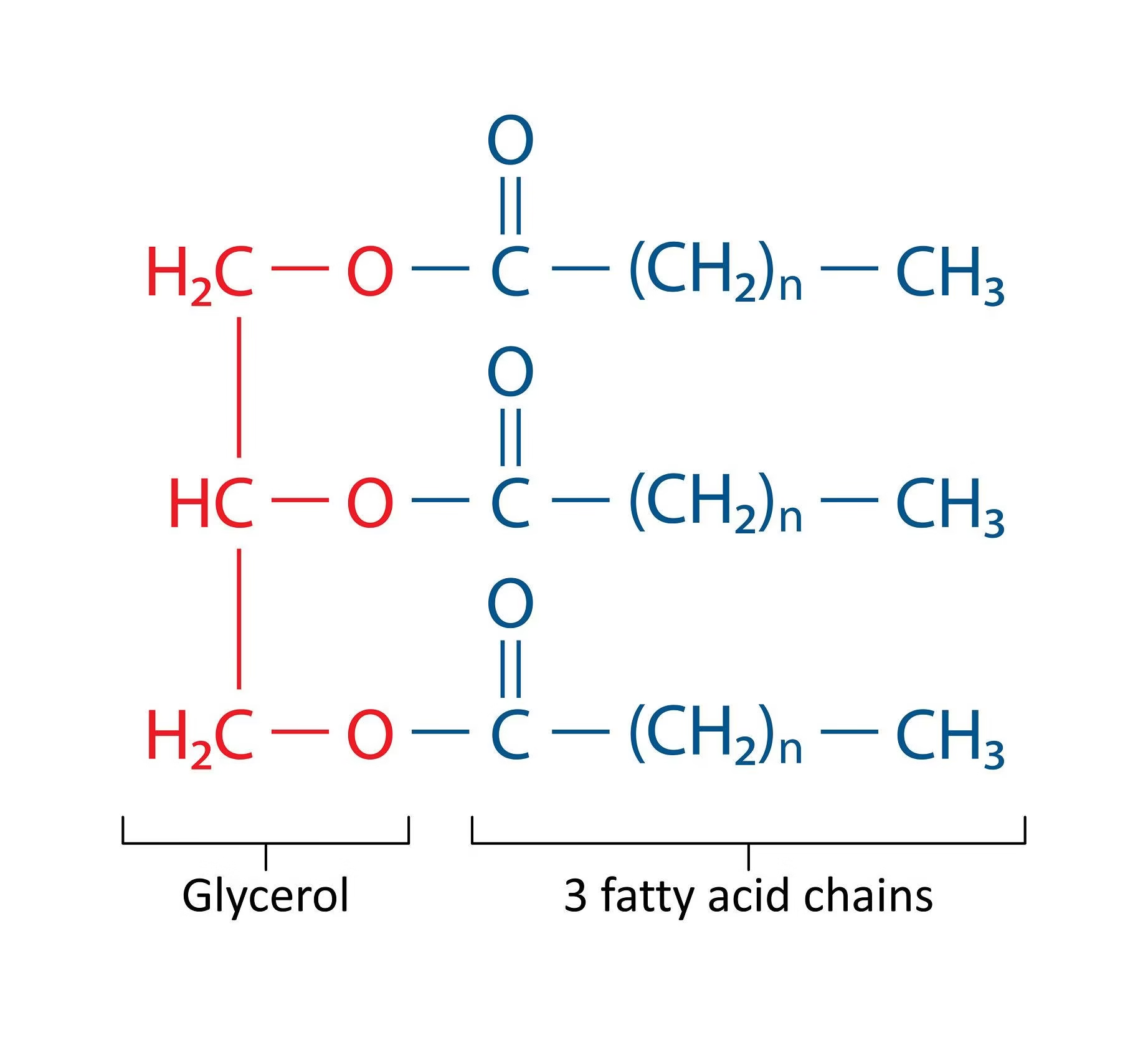
Properties of Triglycerides
-
Triacylglycerols are neutral, non-polar lipids and are completely insoluble in water.
-
They are soluble in non-polar organic solvents such as ether, chloroform, and benzene.
-
They are highly hydrophobic due to the presence of long hydrocarbon chains of fatty acids.
-
They have a high caloric value and yield approximately 9 kcal per gram upon oxidation.
-
Their melting point depends on the length and degree of saturation of fatty acids; saturated triacylglycerols have higher melting points than unsaturated ones.
-
They are lighter than water and have lower density, so they float on the surface of water.
-
They can undergo hydrolysis in the presence of acids, alkali, or lipase enzymes to form glycerol and fatty acids.
-
They are prone to oxidation, especially if they contain unsaturated fatty acids, leading to rancidity.
Functions of Triglycerides
-
Energy storage – Main storage form of energy; provide ~9 kcal/g (more than carbs & proteins).
-
Energy source – Broken down to fatty acids & glycerol for ATP production.
-
Insulation – Subcutaneous fat reduces heat loss, maintaining body temperature.
-
Protection – Cushions and protects vital organs (kidneys, heart, eyes).
-
Metabolic water – Oxidation of triglycerides produces water, important in desert animals.
-
Source of glycerol & fatty acids – For synthesis of phospholipids, glycolipids, and other biomolecules.
Phospholipids
Phospholipids are complex lipids made of glycerol (or sphingosine) + 2 fatty acids + phosphate group + nitrogenous base.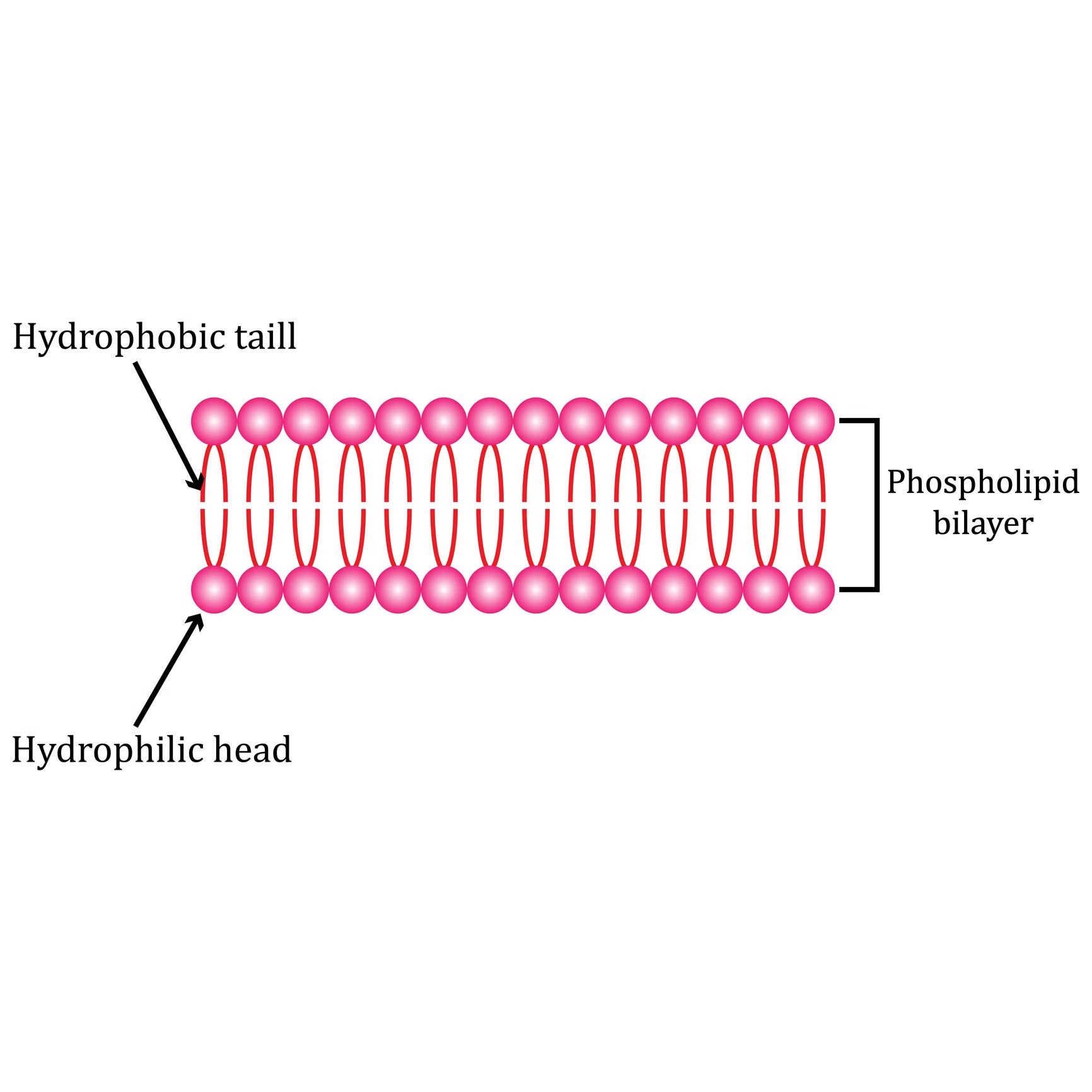
-
They are amphipathic molecules → hydrophilic “head” + hydrophobic “tails”.
-
Major components of cell membranes.
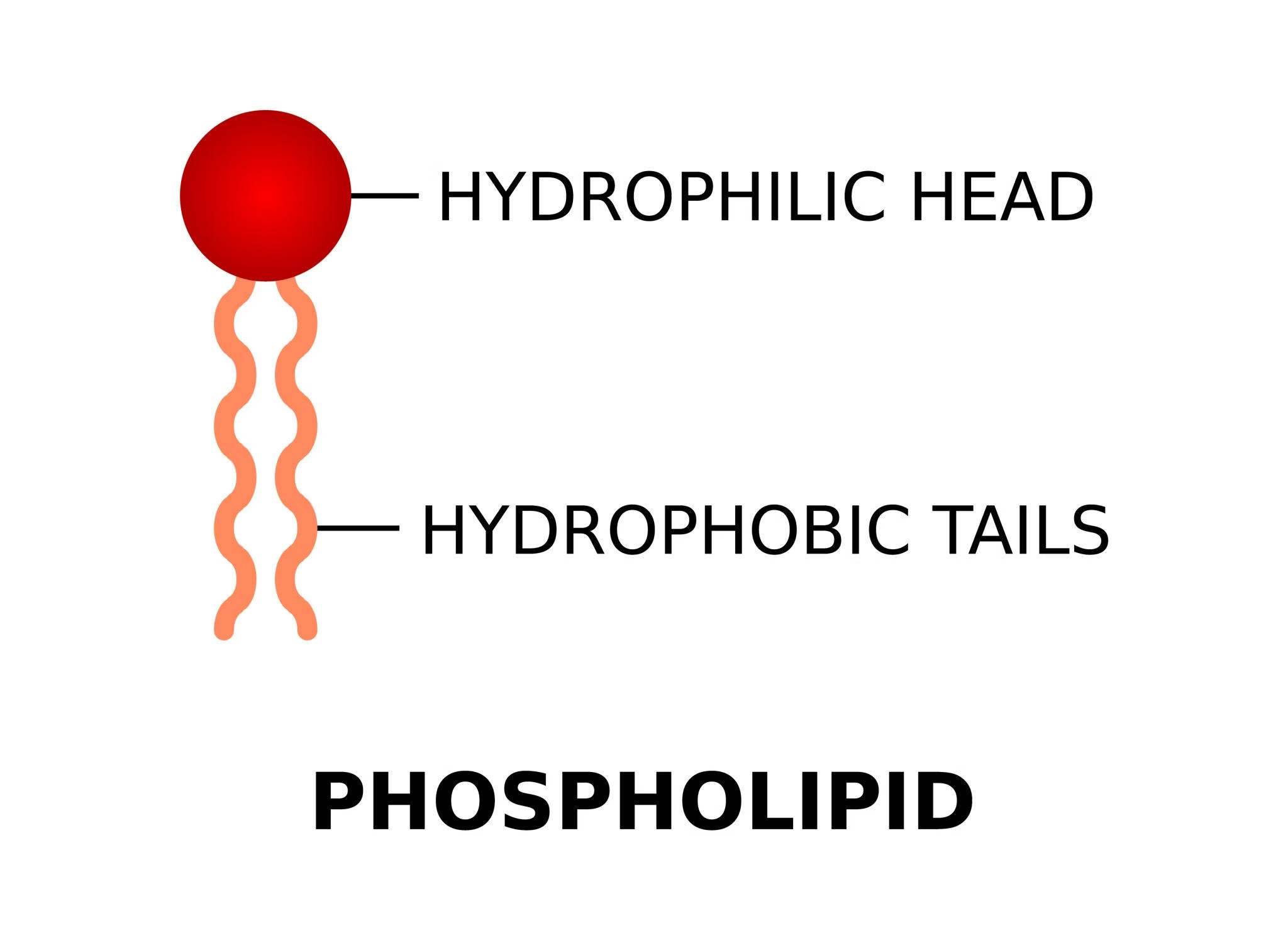
Phospholipids are divided mainly into two groups based on the backbone structure:
1. Glycerophospholipids (Phosphoglycerides)
-
Backbone: Glycerol
-
General structure: Glycerol + 2 fatty acids + phosphate group + nitrogenous base
-
Major types:
-
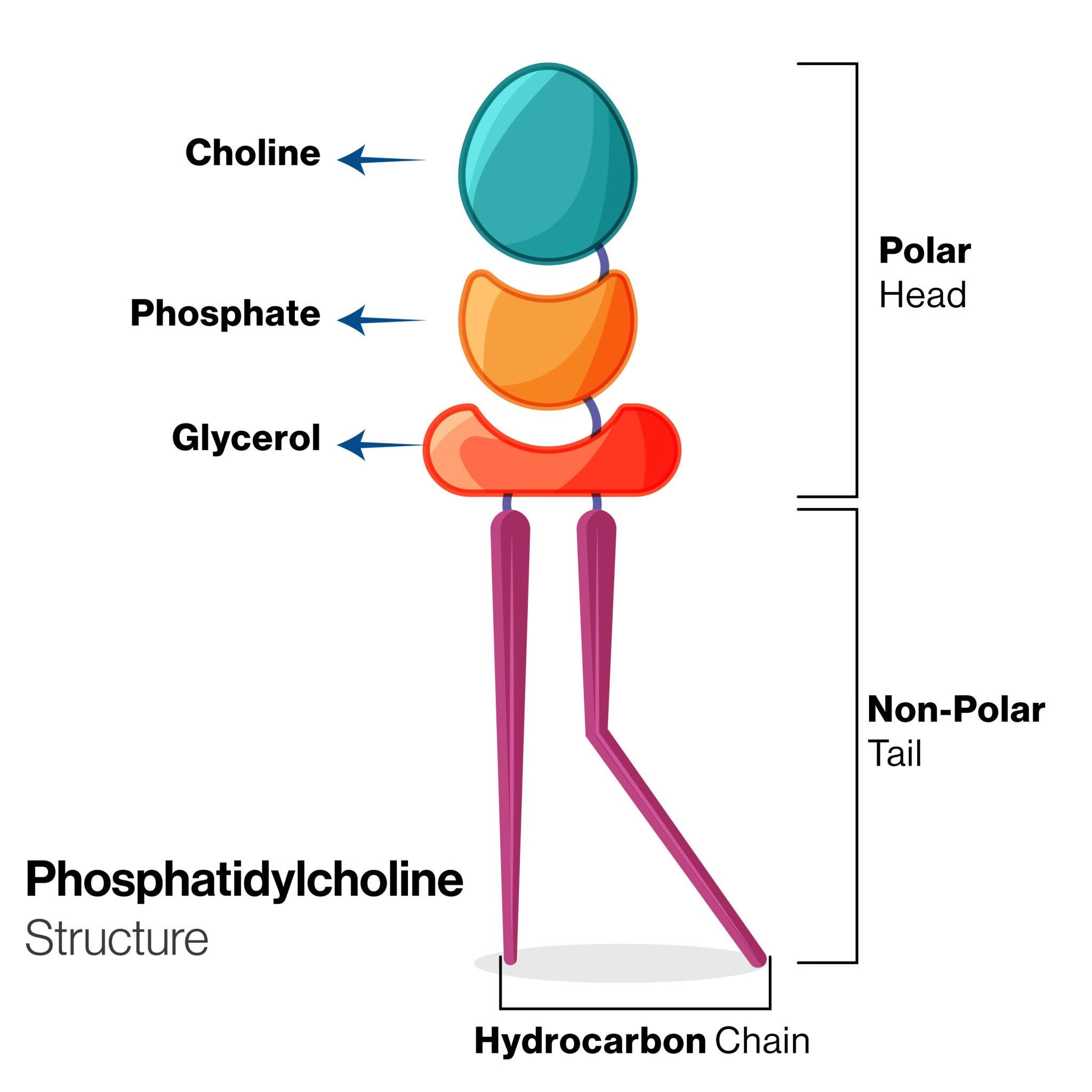
Phosphatidylcholine (Lecithin)
-
Most abundant phospholipid in cell membranes.
-
Found in egg yolk, soybeans, brain tissue.
-
Functions: Structural role, lipid transport, lung surfactant.
-
-
Phosphatidylethanolamine (Cephalin)
-
Found in brain and nervous tissue.
-
Role in membrane structure and blood clotting.
-
-
Phosphatidylserine
-
Found in brain, inner surface of cell membranes.
-
Role in apoptosis (cell death signaling) and blood coagulation.
-
-
Phosphatidylinositol (PI)
-
Contains sugar alcohol inositol.
-
Important in signal transduction (IP₃, DAG pathway) and membrane anchoring.
-
-
Cardiolipin (Diphosphatidylglycerol)
-
Found in inner mitochondrial membrane.
-
Essential for oxidative phosphorylation and energy metabolism.
-
2. Sphingophospholipids
-
Backbone: Sphingosine (instead of glycerol).
-
General structure: Sphingosine + fatty acid + phosphate + choline/ethanolamine.
-
Major type:
Sphingomyelin
-
Found in myelin sheath of nerve fibers.
-
Role: Insulates axons, speeds up nerve impulse transmission.
Functions of Phospholipids
-
Structural role – Major components of cell membranes (lipid bilayer).
-
Membrane fluidity – Maintain the flexibility and permeability of membranes.
-
Signal transduction – Act as precursors of second messengers (e.g., IP₃, DAG).
-
Transport – Form lipoproteins that transport lipids in blood.
-
Surfactant function – Dipalmitoyl lecithin reduces surface tension in lungs (prevents alveolar collapse).
-
Nerve insulation – Sphingomyelin is important in the myelin sheath.
-
Emulsification – Helps in the digestion/absorption of dietary fats with bile.
Glycolipids
-
Glycolipids are complex lipids composed of a lipid moiety covalently linked to one or more carbohydrate residues.
-
They are primarily derived from sphingosine and are commonly classified as glycosphingolipids.
-
Glycolipids are amphipathic in nature, containing a hydrophobic lipid portion and a hydrophilic carbohydrate group.
-
They are predominantly found in the outer leaflet of the plasma membrane, especially in nervous tissue.
-
Glycolipids play an essential role in maintaining membrane stability and structural integrity.
-
They are involved in cell recognition, cell adhesion, and antigenic properties (e.g., blood group antigens).
-
They contribute to signal transduction and interactions between cells and their external environment.
-
Abnormal metabolism of glycolipids can lead to lysosomal storage disorders such as Tay–Sachs disease and Gaucher disease.
Classification of Glycolipids
-
Cerebrosides
-
Structure: Sphingosine + fatty acid + single sugar (glucose or galactose).
-
Example: Galactocerebroside (abundant in myelin sheath).
-
Function: Insulation of nerve fibers.
-
-
Sulfatides (sulfolipids)
-
Cerebrosides with sulfate group attached.
-
Function: Found in brain, kidney → role in cell signaling & myelin stability.
-
-
Globosides
-
Contain two or more sugars (glucose, galactose, N-acetylgalactosamine).
-
Function: Present in cell membranes, involved in cell adhesion & recognition.
-
-
Gangliosides
-
Contain complex oligosaccharides + sialic acid (NANA).
-
Highly abundant in gray matter of brain.
-
Function: Involved in nerve impulse transmission, cell–cell communication, receptor functions.
-
Functions of Glycolipids
-
Structural role – stabilize cell membranes, especially in nerve cells (myelin sheath).
-
Cell recognition & communication – act as markers for cell–cell interactions.
-
Receptors – serve as receptors for toxins, viruses, and hormones (e.g., cholera toxin binds to GM1 ganglioside).
-
Insulation of nerves – cerebrosides & sulfatides maintain myelin sheath.
-
Immune functions – glycolipids on cell surfaces act as antigens (e.g., ABO blood group antigens).
-
Signal transduction – participate in transmitting signals across membranes.
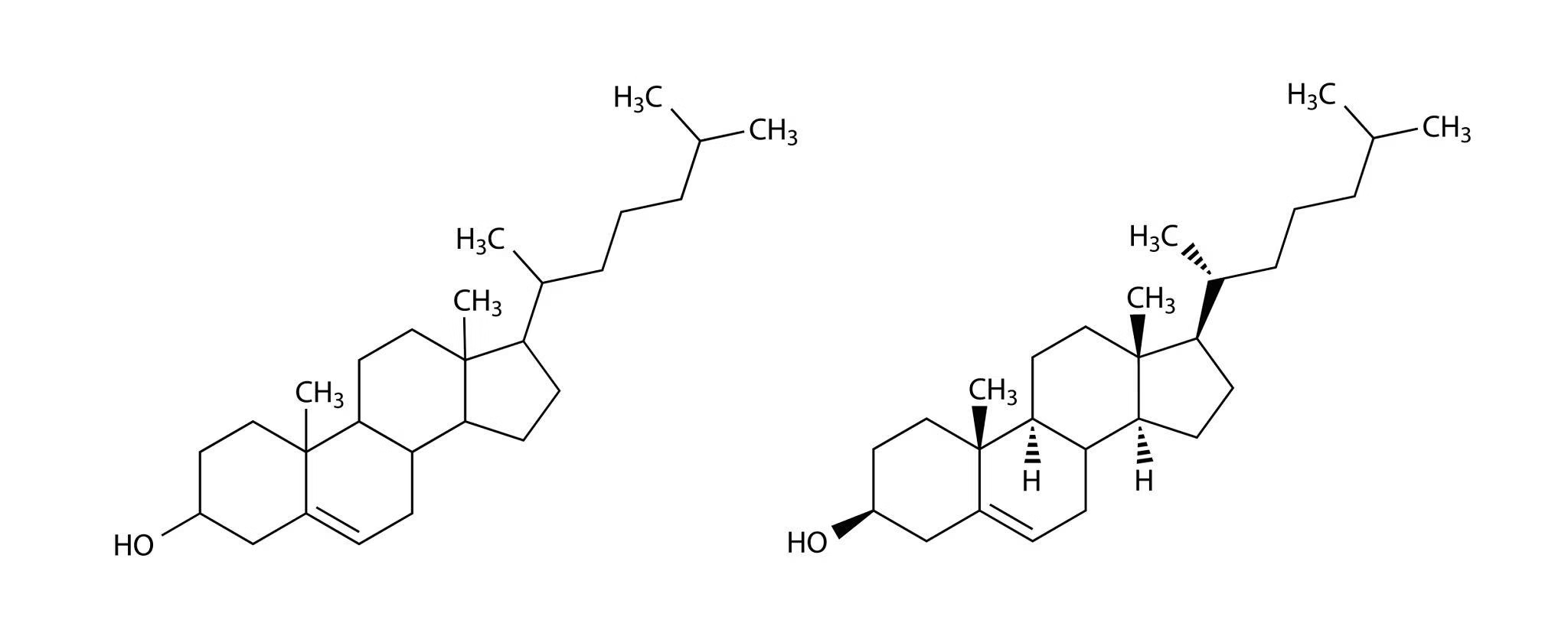
Cholesterol
-
Cholesterol is a sterol (steroid alcohol) and an essential component of animal cell membranes.
-
It contains a characteristic four-ring steroid nucleus with a hydroxyl group, making it amphipathic in nature.
-
Cholesterol is synthesized primarily in the liver from acetyl-CoA through a multi-step biochemical pathway.
-
It plays a crucial role in maintaining membrane fluidity and stability.
-
Cholesterol serves as a precursor for steroid hormones such as glucocorticoids, mineralocorticoids, and sex hormones.
-
It is also the precursor of bile acids, which are necessary for digestion and absorption of dietary lipids.
-
Cholesterol is transported in the bloodstream in association with lipoproteins such as LDL, HDL, and VLDL.
-
Elevated levels of serum cholesterol, particularly low-density lipoprotein (LDL), are associated with an increased risk of atherosclerosis and cardiovascular diseases.
Functions of Cholesterol
1. Structural Role in Cell Membrane – Cholesterol is an essential component of cell membranes, where it maintains membrane fluidity, stability, and permeability.
2. Precursor of Steroid Hormones – It serves as a precursor for steroid hormones such as glucocorticoids, mineralocorticoids, estrogens, progesterone, and testosterone.
3. Formation of Bile Acids and Bile Salts – Cholesterol is converted into bile acids and bile salts, which are necessary for digestion and absorption of dietary lipids.
4. Synthesis of Vitamin D – It acts as a precursor for vitamin D synthesis in the skin under the influence of ultraviolet radiation.
5. Role in Cell Signalling – Cholesterol participates in the formation of lipid rafts, which are specialized membrane domains involved in signal transduction.
6. Nervous System Function – It contributes to the formation and maintenance of the myelin sheath in nerve cells, ensuring proper nerve conduction.
7. Intracellular Transport – Cholesterol plays a role in membrane trafficking and intracellular transport processes.
8. Support of Growth and Development – It is essential for normal cellular growth, differentiation, and embryonic development.
Lipoproteins
-
Lipoproteins are particles made of fats (lipids) and proteins.
-
Their job is to carry fats in the blood, because fats cannot travel alone in water (blood).
-
Structure:
-
Core (inside): contains triglycerides and cholesterol esters (water-insoluble).
-
Outer layer: has phospholipids, free cholesterol, and apolipoproteins (water-soluble).
-
They are classified based on density and function into:
1. Chylomicrons
-
Largest, lowest density lipoproteins.
-
Rich in dietary triglycerides.
-
Synthesized in intestinal mucosa after a meal.
-
Function: Transport dietary triglycerides from intestine → peripheral tissues (for energy/storage). Residue goes to liver.
2. Very Low-Density Lipoproteins (VLDL)
-
Produced by the liver.
-
Rich in endogenous triglycerides (synthesized in liver).
-
Function: Transport triglycerides from liver → peripheral tissues.
-
On metabolism, VLDL is converted → IDL → LDL.
3. Intermediate-Density Lipoproteins (IDL)
-
Transitional form between VLDL and LDL.
-
Contain triglycerides & cholesterol.
-
Function: Carry remaining triglycerides and cholesterol to tissues or liver.
4. Low-Density Lipoproteins (LDL)
-
Formed from VLDL/IDL metabolism.
-
Rich in cholesterol & cholesterol esters.
-
Function: Deliver cholesterol to peripheral tissues for membrane synthesis & hormone production.
-
Excess LDL → deposits in arteries → atherosclerosis (“bad cholesterol”).
5. High-Density Lipoproteins (HDL)
-
-
Smallest, highest density (protein-rich).
-
Synthesized in liver & intestine.
-
Function: Collect excess cholesterol from tissues & blood → transport back to liver (reverse cholesterol transport).
-
Protective role → “good cholesterol.”
-
Functions
-
Transport triglycerides (chylomicrons, VLDL).
-
Carry cholesterol to tissues (LDL = “bad cholesterol”).
-
Remove excess cholesterol back to liver (HDL = “good cholesterol”).
-
Provide energy indirectly by delivering fats to cells.
-
Help in fat absorption from intestine (chylomicrons).
MCQs
-
Lipids are primarily insoluble in:
A. Alcohol
B. Water
C. Ether
D. Chloroform
Answer: B -
Lipids provide how many kcal per gram?
A. 4
B. 7
C. 9
D. 12
Answer: C -
The basic unit of most lipids is:
A. Amino acid
B. Monosaccharide
C. Fatty acid
D. Nucleotide
Answer: C -
Triacylglycerol consists of:
A. 1 fatty acid + 3 glycerol
B. 3 fatty acids + 1 glycerol
C. 2 fatty acids + 2 glycerol
D. 4 fatty acids
Answer: B -
Which lipid is a major component of cell membrane?
A. Triglyceride
B. Phospholipid
C. Wax
D. Cholesteryl ester
Answer: B -
Saturated fatty acids contain:
A. Double bonds
B. Triple bonds
C. No double bonds
D. Peptide bonds
Answer: C -
Oleic acid is a:
A. Saturated fatty acid
B. Monounsaturated fatty acid
C. Polyunsaturated fatty acid
D. Trans fatty acid
Answer: B -
Essential fatty acids must be obtained from:
A. Liver
B. Kidney
C. Diet
D. Muscle
Answer: C -
Linoleic acid belongs to which family?
A. Omega-3
B. Omega-6
C. Omega-9
D. Saturated
Answer: B -
Alpha-linolenic acid is an example of:
A. Omega-3 fatty acid
B. Omega-6 fatty acid
C. Saturated fat
D. Trans fat
Answer: A -
Cholesterol is a:
A. Phospholipid
B. Glycolipid
C. Sterol
D. Triglyceride
Answer: C -
Cholesterol contains:
A. Peptide chain
B. Steroid nucleus
C. Phosphate group
D. Sugar moiety
Answer: B -
Lipids are stored in the body mainly as:
A. Glycogen
B. Cholesterol
C. Triglycerides
D. Phospholipids
Answer: C -
Hydrolysis of triglycerides produces:
A. Glucose
B. Amino acids
C. Glycerol and fatty acids
D. Steroids
Answer: C -
Phospholipids are:
A. Completely hydrophobic
B. Amphipathic
C. Water soluble
D. Proteins
Answer: B -
Rancidity occurs due to:
A. Hydrolysis
B. Oxidation
C. Reduction
D. Polymerization
Answer: B -
Bile acids are derived from:
A. Fatty acids
B. Cholesterol
C. Glycerol
D. Glucose
Answer: B -
Waxes are esters of fatty acids with:
A. Glycerol
B. Long-chain alcohols
C. Amino acids
D. Sugars
Answer: B -
Which lipoprotein is considered “good cholesterol”?
A. LDL
B. HDL
C. VLDL
D. Chylomicron
Answer: B -
LDL mainly transports:
A. Triglycerides
B. Cholesterol to tissues
C. Amino acids
D. Glucose
Answer: B -
Glycolipids are abundant in:
A. Liver
B. Muscle
C. Nervous tissue
D. Bone
Answer: C -
Prostaglandins are derived from:
A. Amino acids
B. Cholesterol
C. Essential fatty acids
D. Glucose
Answer: C -
The backbone of triglyceride is:
A. Sphingosine
B. Glycerol
C. Cholesterol
D. Glucose
Answer: B -
Saponification involves:
A. Oxidation
B. Reduction
C. Alkaline hydrolysis
D. Hydrogenation
Answer: C -
Hydrogenation converts:
A. Saturated fats to unsaturated
B. Unsaturated fats to saturated
C. Fats to proteins
D. Lipids to glucose
Answer: B -
Trans fats are formed during:
A. Hydrolysis
B. Hydrogenation
C. Oxidation
D. Digestion
Answer: B -
Essential fatty acid deficiency causes:
A. Night blindness
B. Dermatitis
C. Scurvy
D. Anemia
Answer: B -
Lipids float on water because they are:
A. Heavy
B. Polar
C. Less dense
D. Charged
Answer: C -
The major storage site of lipids is:
A. Brain
B. Adipose tissue
C. Kidney
D. Pancreas
Answer: B -
Fatty acids usually contain:
A. Odd number of carbons
B. Even number of carbons
C. Nitrogen
D. Sulfur
Answer: B -
The carboxyl group in fatty acids is:
A. –NH₂
B. –COOH
C. –OH
D. –CH₃
Answer: B -
Lipid digestion primarily occurs in the:
A. Mouth
B. Stomach
C. Small intestine
D. Large intestine
Answer: C -
Lipase enzyme breaks down:
A. Proteins
B. Carbohydrates
C. Lipids
D. Nucleic acids
Answer: C -
Omega numbering counts from the:
A. Carboxyl end
B. Methyl end
C. Middle
D. Alpha carbon
Answer: B -
Myelin sheath is rich in:
A. Proteins
B. Lipids
C. Carbohydrates
D. Nucleic acids
Answer: B -
Which vitamin is fat-soluble?
A. Vitamin C
B. Vitamin B12
C. Vitamin A
D. Vitamin B6
Answer: C -
VLDL mainly transports:
A. Cholesterol from liver to tissues
B. Triglycerides from liver
C. Amino acids
D. Oxygen
Answer: B -
Chylomicrons transport dietary:
A. Glucose
B. Amino acids
C. Triglycerides
D. Urea
Answer: C -
The amphipathic nature is essential for:
A. Energy storage
B. Membrane formation
C. Insulation
D. Protection
Answer: B -
The steroid nucleus has:
A. 2 rings
B. 3 rings
C. 4 rings
D. 5 rings
Answer: C -
Saturated fats are generally:
A. Liquid at room temperature
B. Solid at room temperature
C. Gas
D. Polar
Answer: B -
Unsaturated fats have:
A. Only single bonds
B. One or more double bonds
C. Peptide bonds
D. Phosphate bonds
Answer: B -
Cholesterol synthesis occurs mainly in the:
A. Brain
B. Kidney
C. Liver
D. Spleen
Answer: C -
Lipid oxidation produces:
A. Energy
B. Nitrogen
C. Peptides
D. Vitamins
Answer: A -
Eicosanoids are derived from:
A. Arachidonic acid
B. Glucose
C. Cholesterol
D. Glycerol
Answer: A -
Glycosphingolipids contain:
A. Glycerol
B. Sphingosine
C. Peptide
D. DNA
Answer: B -
Lipid peroxidation affects:
A. DNA replication
B. Membrane integrity
C. Protein synthesis
D. Glycolysis
Answer: B -
High LDL increases risk of:
A. Diabetes
B. Hypertension
C. Atherosclerosis
D. Anemia
Answer: C -
Ketone bodies are produced during:
A. Excess carbohydrate intake
B. Fat metabolism
C. Protein synthesis
D. Vitamin deficiency
Answer: B -
The simplest lipids are:
A. Glycolipids
B. Phospholipids
C. Simple lipids
D. Lipoproteins
Answer: C

