Introduction
-
The LE (Lupus Erythematosus) cell phenomenon is a classical immuno-hematological finding associated with autoimmune diseases.
-
It was first described in 1948 by Hargraves et al., marking a major milestone in the understanding of autoimmunity.
-
The phenomenon is most commonly linked with systemic lupus erythematosus (SLE).
-
LE cell formation represents an antibody-mediated immune complex reaction involving nuclear material.
-
It demonstrates the presence of antinuclear antibodies (ANA) directed against nuclear components.
-
The LE cell itself is a normal phagocytic cell (neutrophil or macrophage), not a diseased cell.
-
The ingested material is a hematoxylin body, derived from damaged or denatured cell nuclei.
-
Historically, the LE cell test was one of the earliest laboratory tests used for diagnosing SLE.
-
Due to low sensitivity and specificity, it has been replaced by modern immunological assays.
-
At present, the LE cell phenomenon is of academic and teaching importance, helping in understanding the pathogenesis of autoimmune disorders.
Historical Background
-
First described in 1948 by Hargraves et al.
-
Provided the first laboratory proof of an autoimmune mechanism in SLE
-
Marked the beginning of autoimmune diagnostics in clinical medicine
Definition of LE Cell
An LE (Lupus Erythematosus) cell is a neutrophil or macrophage that has phagocytosed denatured nuclear material, known as a hematoxylin body (LE body), which is coated with antinuclear antibodies (ANA).
Key point:
The LE cell itself is not diseased. It is a normal phagocytic cell responding to abnormal immune complexes formed due to autoantibody-mediated nuclear damage.
Hematoxylin Body
The hematoxylin body, also called the LE body, represents the altered nuclear material that triggers LE cell formation.
Origin
-
Derived from damaged or apoptotic cell nuclei
-
Nuclear components (DNA, histones, nucleoproteins) become exposed and antibody-coated
Morphological Features
The hematoxylin body characteristically appears as:
-
Homogeneous
-
Round or oval
-
Smooth and structureless
-
Basophilic (deep purple–blue on Wright/Giemsa or hematoxylin staining)
-
Loss of nuclear chromatin details (no visible nucleoli or chromatin pattern)
Microscopic Significance
-
Represents denatured nuclear material
-
Central diagnostic component of the LE cell
-
Distinguishes LE cells from tart cells (which contain intact nuclei)
Pathogenesis of LE Cell Formation
Step-by-Step Pathogenesis
1. Cellular Injury and Nuclear Material Release
-
Increased apoptosis or cell damage occurs in SLE.
-
Defective clearance of apoptotic cells leads to persistence of nuclear debris (DNA, histones, nucleoproteins) in extracellular spaces.
2. Formation of Antinuclear Antibodies (ANA)
-
Loss of self-tolerance results in production of ANA directed against:
-
Double-stranded DNA
-
Histones
-
Nucleoproteins
-
-
These antibodies circulate freely in patient serum.
3. Antigen–Antibody Complex Formation
-
ANA bind to exposed nuclear antigens released from damaged cells.
-
This binding produces immune complexes composed of:
-
Nuclear material
-
Immunoglobulins (mainly IgG)
-
4. Complement Activation and Opsonization
-
Immune complexes activate the classical complement pathway.
-
Complement components (C3b) coat the complexes, enhancing opsonization.
5. Conversion into Hematoxylin Body (LE Body)
-
Bound antibodies cause denaturation and homogenization of nuclear material.
-
Nuclear chromatin loses structure and forms a:
-
Smooth
-
Round
-
Homogeneous
basophilic mass called the hematoxylin body.
-
6. Phagocytosis by Neutrophils or Macrophages
-
Opsonized hematoxylin bodies are recognized by:
-
Fc receptors
-
Complement receptors
-
-
Neutrophils (most common) or macrophages engulf the hematoxylin body.
7. Formation of the LE Cell
-
The phagocyte containing the ingested hematoxylin body becomes an LE cell.
-
The cell’s own nucleus is:
-
Pushed to the periphery
-
Crescent-shaped or flattened
-

Methods of Demonstration of LE Cell Phenomenon
Peripheral Blood Smear Method
Principle
In patients with systemic lupus erythematosus (SLE), antinuclear antibodies (ANA) present in the blood react with exposed nuclear material, leading to the formation of hematoxylin bodies, which may be phagocytosed by neutrophils or monocytes and visualized directly in a peripheral blood smear.
Procedure
-
Collect fresh venous blood from the patient
-
Prepare a thin peripheral blood smear on a clean glass slide
-
Allow the smear to air-dry
-
Stain using:
-
Wright stain or
-
Giemsa stain
-
-
Examine the smear under:
-
Low power (screening)
-
Oil immersion (confirmation)
-
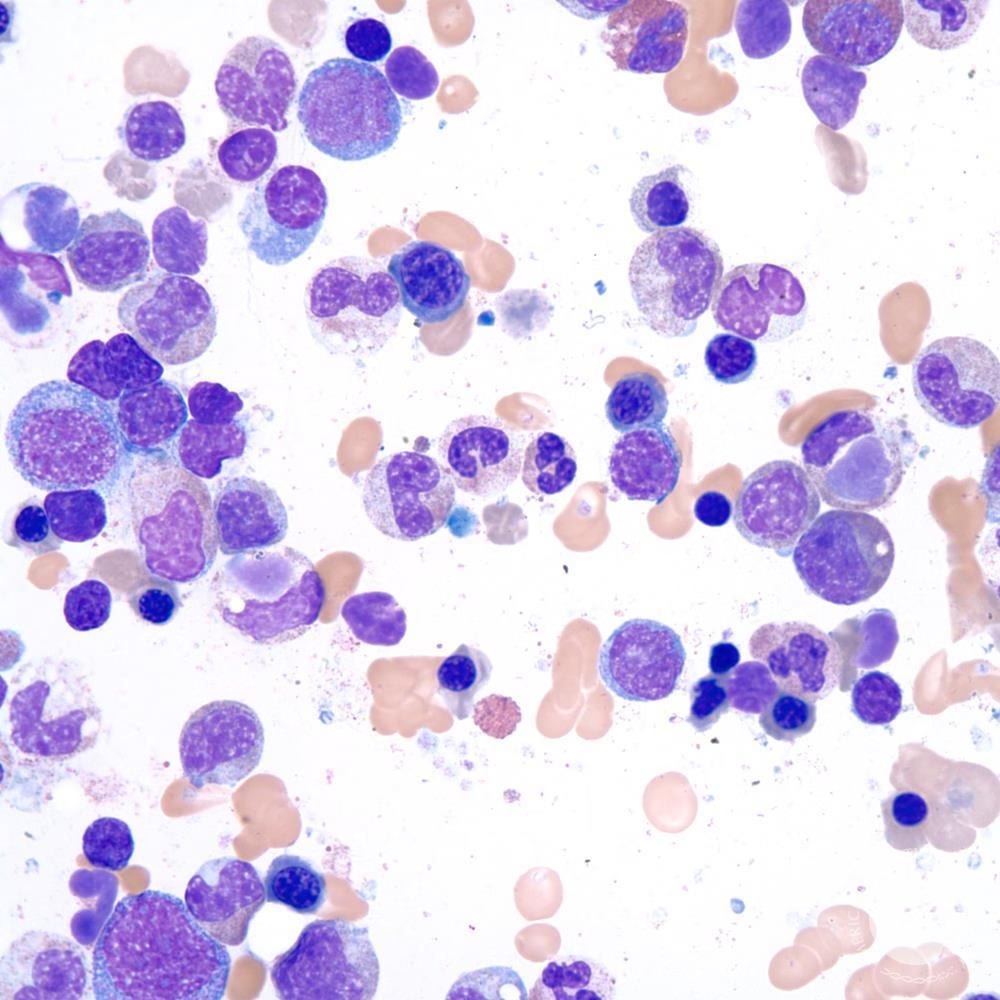
Microscopic Findings
-
Occasional LE cells may be seen
-
Typical features:
-
Neutrophil containing a round, homogeneous, basophilic hematoxylin body
-
Cell’s own nucleus displaced to the periphery
-
-
LE cells are scanty and infrequent
Interpretation
| Finding | Significance |
|---|---|
| Presence of LE cell | Suggestive of autoimmune activity |
| Absence of LE cell | Does not rule out SLE |
Advantages
-
Simple and rapid
-
No special equipment required
-
Can be done in routine hematology labs
Limitations
-
Very low sensitivity
-
LE cells are rarely present
-
High false-negative rate
-
Requires active disease for positivity
-
Not suitable as a screening test

Buffy Coat Technique
Principle
- When whole blood from a patient with systemic lupus erythematosus (SLE) is allowed to clot and incubate, nuclear material is released from damaged leukocytes.
- Antinuclear antibodies (ANA) present in the serum bind to this nuclear material, forming immune complexes that are subsequently phagocytosed by neutrophils, resulting in LE cell formation.
- The buffy coat, being rich in leukocytes, increases the likelihood of detecting LE cells.
Procedure
-
Collect 5–10 mL of venous blood in a plain tube (without anticoagulant)
-
Allow the blood to clot at room temperature
-
Mechanically disrupt the clot using a glass rod to release nuclear material
-
Incubate the disrupted clot at 37°C for 1–2 hours
-
Centrifuge the sample
-
Carefully collect the buffy coat layer (between plasma and RBCs)
-
Prepare a smear from the buffy coat
-
Air-dry and stain using:
-
Wright stain or
-
Giemsa stain
-
-
Examine under oil immersion microscopy
Microscopic Findings
-
Numerous LE cells may be seen
-
Typical LE cell features:
-
Neutrophil or macrophage
-
Large, homogeneous, basophilic hematoxylin body
-
Phagocyte nucleus displaced to the periphery
-
Advantages
-
Higher sensitivity than peripheral blood smear
-
Classical and well-established method
-
Requires no advanced immunological equipment
-
Useful for demonstration and teaching
Limitations
-
Time-consuming
-
Requires careful handling and incubation
-
Low specificity
-
False negatives may still occur
-
Obsolete in routine clinical practice
Bone Marrow Smear Examination
Principle
- In patients with systemic lupus erythematosus (SLE), antinuclear antibodies (ANA) bind to nuclear material released from damaged cells.
- Within the bone marrow environment, this antibody-coated nuclear material is efficiently phagocytosed by macrophages or neutrophils, leading to LE cell formation, which can be directly visualized in marrow smears.
Procedure
-
Perform bone marrow aspiration (usually from:
-
Posterior superior iliac spine
-
Sternum – rarely)
-
-
Prepare thin bone marrow smears on clean glass slides
-
Allow smears to air-dry
-
Stain using:
-
Wright stain
-
Giemsa stain
-
Leishman stain
-
-
Examine under:
-
Low power (screening)
-
Oil immersion (confirmation)
-
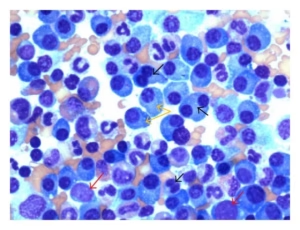
Microscopic Findings
-
LE cells may be present in significant numbers
-
Commonly seen as:
-
Macrophages containing a large hematoxylin body
-
Homogeneous, round, basophilic inclusion
-
-
The phagocyte’s own nucleus is:
-
Flattened
-
Pushed to the periphery
-
Advantages
-
Higher yield of LE cells compared to peripheral blood
-
Clear demonstration of phagocytosis
-
Useful when marrow examination is already indicated
Limitations
-
Invasive procedure
-
Not ethically justified solely for LE cell detection
-
Requires skilled personnel
-
Obsolete as a diagnostic tool for SLE
Clot Incubation Method
Principle
When patient blood is allowed to clot and incubated, leukocyte disruption releases nuclear material. Antinuclear antibodies (ANA) present in the serum bind this material, forming immune complexes that are subsequently phagocytosed by neutrophils, producing LE cells.
Procedure
-
Collect venous blood in a plain tube (no anticoagulant).
-
Allow blood to clot at room temperature.
-
Disrupt the clot gently with a glass rod to expose nuclear material.
-
Incubate at 37 °C for 1–2 hours.
-
Prepare smears from the disrupted clot.
-
Stain with Wright/Giemsa and examine under oil immersion.
Microscopic Findings
-
Increased number of LE cells compared with direct peripheral smear.
-
Neutrophils containing a homogeneous, basophilic hematoxylin body with peripheral displacement of the cell’s own nucleus.
Merits and Limitations
-
Merit: Better sensitivity than peripheral smear; simple setup.
-
Limitations: Time-consuming; still low specificity; largely obsolete.
Indirect LE Cell Test
Principle
This method demonstrates the antibody-mediated mechanism of LE cell formation. Patient serum (containing ANA) is incubated with normal donor leukocytes. If ANA are present, they bind exposed nuclear material and induce LE cell formation in otherwise normal cells.
Procedure
-
Collect serum from the patient (suspected SLE).
-
Obtain normal leukocytes from a healthy donor.
-
Mix patient serum with donor leukocytes.
-
Incubate at 37 °C.
-
Prepare smears and stain with Wright/Giemsa.
Interpretation
-
Positive test: Formation of LE cells → presence of circulating ANA.
-
Negative test: No LE cells → ANA absent or below detectable level.
Significance
-
Proves that LE cell formation is serum (antibody) dependent.
-
Research and teaching value; not used for routine diagnosis.
Diagnostic Significance
1. Association with Diseases
| Disease | Diagnostic Relevance |
|---|---|
| Systemic lupus erythematosus (SLE) | Classically associated |
| Rheumatoid arthritis | Occasional |
| Scleroderma | Rare |
| Dermatomyositis | Rare |
| Drug-induced lupus | Sometimes |
LE cells are most frequently detected in active SLE.
2. Indicator of Autoimmune Activity
-
Demonstrates presence of antinuclear antibodies (ANA)
-
Reflects immune complex formation
-
Indicates ongoing nuclear damage and defective clearance
-
Suggests active autoimmune process
3. Sensitivity and Specificity
| Parameter | Diagnostic Value |
|---|---|
| Sensitivity | Low to moderate |
| Specificity | Low |
| Screening utility | Poor |
| Confirmatory role | None |
Absence of LE cells does not exclude SLE.
4. Correlation with Disease Activity
-
More likely to be positive during active disease
-
Less frequently detected during remission
-
Rough correlation with disease severity
5. Reasons for Obsolescence
The LE cell test has been replaced by modern immunological assays that offer better accuracy:
| Modern Test | Advantage |
|---|---|
| ANA (IIF) | High sensitivity |
| Anti-dsDNA | High specificity |
| Anti-Sm | Diagnostic specificity |
| Complement levels | Disease activity monitoring |
MCQs
1. LE cell phenomenon is most commonly associated with:
A. Rheumatoid arthritis
B. Systemic lupus erythematosus
C. Scleroderma
D. Dermatomyositis
✅ Answer: B
2. An LE cell is best defined as:
A. A diseased neutrophil
B. A macrophage undergoing apoptosis
C. A phagocyte containing denatured nuclear material
D. A lymphocyte with nuclear damage
✅ Answer: C
3. The denatured nuclear material ingested by an LE cell is called:
A. Inclusion body
B. Howell–Jolly body
C. Hematoxylin body
D. Döhle body
✅ Answer: C
4. Hematoxylin bodies are derived from:
A. Cytoplasmic proteins
B. Mitochondria
C. Damaged cell nuclei
D. Ribosomes
✅ Answer: C
5. The antibodies responsible for LE cell formation are:
A. Anti-mitochondrial antibodies
B. Antiphospholipid antibodies
C. Antinuclear antibodies
D. Anti-CCP antibodies
✅ Answer: C
6. Most common cell forming an LE cell is:
A. Lymphocyte
B. Eosinophil
C. Neutrophil
D. Basophil
✅ Answer: C
7. LE cell phenomenon primarily represents:
A. Cell-mediated immunity
B. Immune deficiency
C. Antibody-mediated autoimmunity
D. Hypersensitivity type I
✅ Answer: C
8. Chromatin appearance in hematoxylin body is:
A. Coarse and clumped
B. Intact and segmented
C. Homogeneous and structureless
D. Reticular
✅ Answer: C
9. Stain commonly used to demonstrate LE cells:
A. Ziehl–Neelsen
B. Gram stain
C. Wright–Giemsa
D. PAS
✅ Answer: C
10. Peripheral blood smear method for LE cells has:
A. High sensitivity
B. High specificity
C. Low sensitivity
D. High predictive value
✅ Answer: C
11. Most sensitive classical method for LE cell detection:
A. Peripheral smear
B. Buffy coat technique
C. Bone marrow biopsy
D. Serum electrophoresis
✅ Answer: B
12. Buffy coat is rich in:
A. Platelets only
B. Plasma proteins
C. Leukocytes
D. Red cells
✅ Answer: C
13. Anticoagulant used in buffy coat method:
A. EDTA
B. Heparin
C. Citrate
D. None
✅ Answer: D
14. Incubation temperature for buffy coat technique:
A. 4°C
B. 25°C
C. 37°C
D. 56°C
✅ Answer: C
15. Bone marrow LE cells are commonly seen in:
A. Lymphocytes
B. Plasma cells
C. Macrophages
D. Megakaryocytes
✅ Answer: C
16. Bone marrow examination for LE cells is:
A. Routine
B. Non-invasive
C. Highly specific
D. Rarely justified
✅ Answer: D
17. Clot incubation method improves LE cell detection by:
A. Increasing antibody synthesis
B. Releasing nuclear material
C. Increasing RBC count
D. Enhancing staining
✅ Answer: B
18. Indirect LE cell test demonstrates:
A. Complement deficiency
B. Cellular immunity
C. Antibody-dependent mechanism
D. Genetic defect
✅ Answer: C
19. In indirect LE cell test, patient serum is mixed with:
A. Patient RBCs
B. Normal donor leukocytes
C. Bone marrow cells
D. Plasma proteins
✅ Answer: B
20. Positive indirect LE cell test indicates presence of:
A. Anti-dsDNA only
B. Antinuclear antibodies
C. Rheumatoid factor
D. Anti-CCP
✅ Answer: B
21. LE cell nucleus of phagocyte is usually:
A. Enlarged
B. Destroyed
C. Displaced peripherally
D. Multilobed centrally
✅ Answer: C
22. LE cell phenomenon correlates best with:
A. Age of patient
B. Disease remission
C. Disease activity
D. Gender
✅ Answer: C
23. LE cells are least commonly seen in:
A. SLE
B. Drug-induced lupus
C. Rheumatoid arthritis
D. Iron deficiency anemia
✅ Answer: D
24. Tart cell differs from LE cell because it:
A. Contains homogeneous nucleus
B. Has antibody-coated material
C. Contains intact nucleus
D. Is specific for SLE
✅ Answer: C
25. LE cell test specificity is:
A. Very high
B. Moderate
C. Low
D. Absolute
✅ Answer: C
26. LE cell test sensitivity is:
A. Very high
B. Moderate to low
C. Absolute
D. 100%
✅ Answer: B
27. LE cell phenomenon was first described in:
A. 1920
B. 1935
C. 1948
D. 1965
✅ Answer: C
28. LE cell test is currently:
A. Gold standard
B. Screening test
C. Confirmatory test
D. Obsolete
✅ Answer: D
29. Best modern replacement of LE cell test:
A. ESR
B. CRP
C. ANA by immunofluorescence
D. Widal test
✅ Answer: C
30. Immune complexes in LE cell formation activate:
A. Alternative complement pathway
B. Lectin pathway
C. Classical complement pathway
D. Coagulation pathway
✅ Answer: C
31. LE cell phenomenon demonstrates loss of:
A. Immune surveillance
B. Self-tolerance
C. Phagocytic function
D. Cytokine regulation
✅ Answer: B
32. Hematoxylin body staining color is:
A. Eosinophilic
B. Pale pink
C. Basophilic
D. Colorless
✅ Answer: C
33. LE cell formation requires:
A. Only leukocytes
B. Only ANA
C. Nuclear material + ANA
D. Platelets
✅ Answer: C
34. Which is NOT a method of LE cell demonstration?
A. Buffy coat technique
B. Clot incubation method
C. Indirect serum transfer
D. ELISA
✅ Answer: D
35. LE cells are most easily demonstrated in:
A. Urine sediment
B. CSF
C. Blood/bone marrow smears
D. Saliva
✅ Answer: C
36. LE cell test is mainly useful today for:
A. Patient management
B. Prognosis
C. Teaching and exams
D. Screening
✅ Answer: C
37. The LE cell itself is:
A. Malignant
B. Infected
C. Normal phagocyte
D. Degenerated
✅ Answer: C
38. Loss of chromatin detail in hematoxylin body is due to:
A. Necrosis
B. Enzymatic digestion
C. Antibody-mediated denaturation
D. Staining artifact
✅ Answer: C
39. LE cells are usually absent in:
A. Active SLE
B. Remission phase
C. Drug-induced lupus
D. Autoimmune disease
✅ Answer: B
40. Which antibody is most directly involved in LE cell formation?
A. IgA
B. IgE
C. IgG
D. IgM
✅ Answer: C
41. The buffy coat layer lies between:
A. Plasma and serum
B. Plasma and RBCs
C. RBCs and platelets
D. Serum and clot
✅ Answer: B
42. LE cell phenomenon is an example of:
A. Type I hypersensitivity
B. Type II hypersensitivity
C. Type III hypersensitivity
D. Type IV hypersensitivity
✅ Answer: C
43. Which finding best distinguishes LE cell from Tart cell?
A. Cell size
B. Antibody coating
C. Cytoplasmic granules
D. Cell membrane
✅ Answer: B
44. LE cell test false negatives are:
A. Rare
B. Impossible
C. Common
D. Diagnostic
✅ Answer: C
45. LE cell phenomenon demonstrates:
A. Defective RBC production
B. Immune complex phagocytosis
C. Complement deficiency
D. Enzyme deficiency
✅ Answer: B
46. LE cell test should NOT be used as:
A. Teaching tool
B. Historical reference
C. Screening test
D. Viva topic
✅ Answer: C
47. Which cell is least likely to form LE cell?
A. Neutrophil
B. Macrophage
C. Monocyte
D. Erythrocyte
✅ Answer: D
48. Nuclear material in LE cell appears:
A. Segmented
B. Granular
C. Homogeneous
D. Vacuolated
✅ Answer: C
49. LE cell phenomenon helped establish concept of:
A. Infection
B. Malignancy
C. Autoimmune disease
D. Genetic disorder
✅ Answer: C
50. Final conclusion about LE cell test:
A. Still gold standard
B. Highly specific
C. Clinically obsolete but educationally important
D. Diagnostic of SLE
✅ Answer: C
