Introduction
-
Hemostasis is the physiological process that prevents excessive blood loss following injury to a blood vessel.
-
It maintains a delicate balance between clot formation and blood fluidity in the circulation.
-
The hemostatic mechanism involves the coordinated action of blood vessels, platelets, coagulation factors, and the fibrinolytic system.
-
It is a step-wise and highly regulated process, not a single event.
-
Proper functioning of hemostasis is essential for normal wound healing and vascular integrity.
-
Disturbances in this mechanism can lead to bleeding disorders or pathological thrombosis.
-
Understanding the hemostatic mechanism is crucial for students of medical biochemistry, hematology, and clinical medicine.
What is Hemostasis?
Hemostasis means:
-
Stopping blood loss at the site of injury
-
Forming a clot only where needed
-
Removing the clot after healing
In simple words, it is the body’s emergency repair system for blood vessels.
Clotting factors
Clotting factors are plasma proteins (mostly synthesized in the liver) that interact in a cascade mechanism to achieve hemostasis by forming a stable fibrin clot.
| Factor No. | Name | Major Function in Hemostasis |
|---|---|---|
| Factor I | Fibrinogen | Converted to fibrin by thrombin; forms the structural framework of the clot |
| Factor II | Prothrombin | Converted to thrombin, which converts fibrinogen to fibrin and activates other factors |
| Factor III | Tissue Factor (Thromboplastin) | Initiates extrinsic pathway by activating factor VII |
| Factor IV | Calcium (Ca²⁺) | Essential cofactor for activation of multiple clotting factors |
| Factor V | Proaccelerin (Labile factor) | Cofactor for factor Xa in conversion of prothrombin to thrombin |
| Factor VII | Proconvertin | Activates extrinsic pathway; activates factor X |
| Factor VIII | Anti-hemophilic factor A | Cofactor for factor IXa; deficiency causes Hemophilia A |
| Factor IX | Christmas factor | Activates factor X in intrinsic pathway; deficiency causes Hemophilia B |
| Factor X | Stuart–Prower factor | Converts prothrombin to thrombin (common pathway) |
| Factor XI | Plasma thromboplastin antecedent | Activates factor IX in intrinsic pathway |
| Factor XII | Hageman factor | Initiates intrinsic pathway and activates fibrinolysis |
| Factor XIII | Fibrin-stabilizing factor | Cross-links fibrin → stable clot |
| von Willebrand Factor (vWF) | — | Mediates platelet adhesion and stabilizes factor VIII |
Components of Hemostatic Mechanism
Hemostasis depends on four main components:
-
Blood vessels (endothelium)
-
Platelets
-
Coagulation factors (clotting factors)
-
Fibrinolytic system (clot removal system)
All these work together in a fixed sequence.
Steps of Hemostatic Mechanism
Step 1: Vascular Spasm (Immediate Response)
-
As soon as a blood vessel is injured, it contracts (vasoconstriction)
-
This reduces blood flow and minimizes blood loss
How does it occur?
-
Release of endothelin from damaged endothelium
-
Local nerve reflexes
This step is temporary but very important.
Step 2: Primary Hemostasis (Platelet Plug Formation) 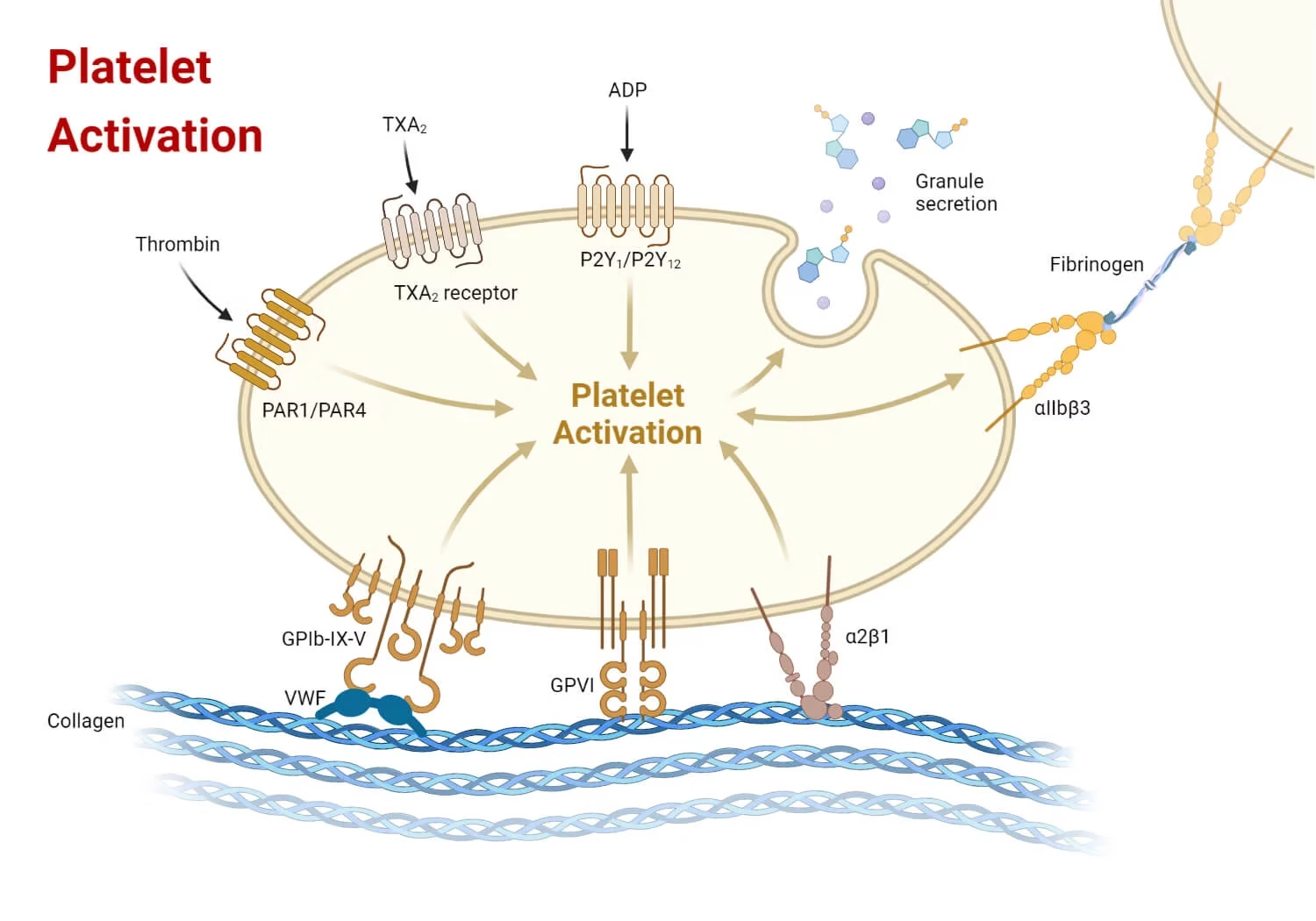
This step involves platelets.
2.1 Platelet Adhesion
-
Injury exposes collagen in the vessel wall
-
Platelets stick to collagen with the help of von Willebrand factor (vWF)
2.2 Platelet Activation
-
Adhered platelets become activated and release chemicals:
-
ADP
-
Serotonin
-
Thromboxane A₂
-
These chemicals:
-
Attract more platelets
-
Increase platelet stickiness
2.3 Platelet Aggregation
-
Platelets stick to each other
-
Form a temporary platelet plug
This plug is weak and unstable, so it needs reinforcement.
Step 3: Secondary Hemostasis (Coagulation Cascade)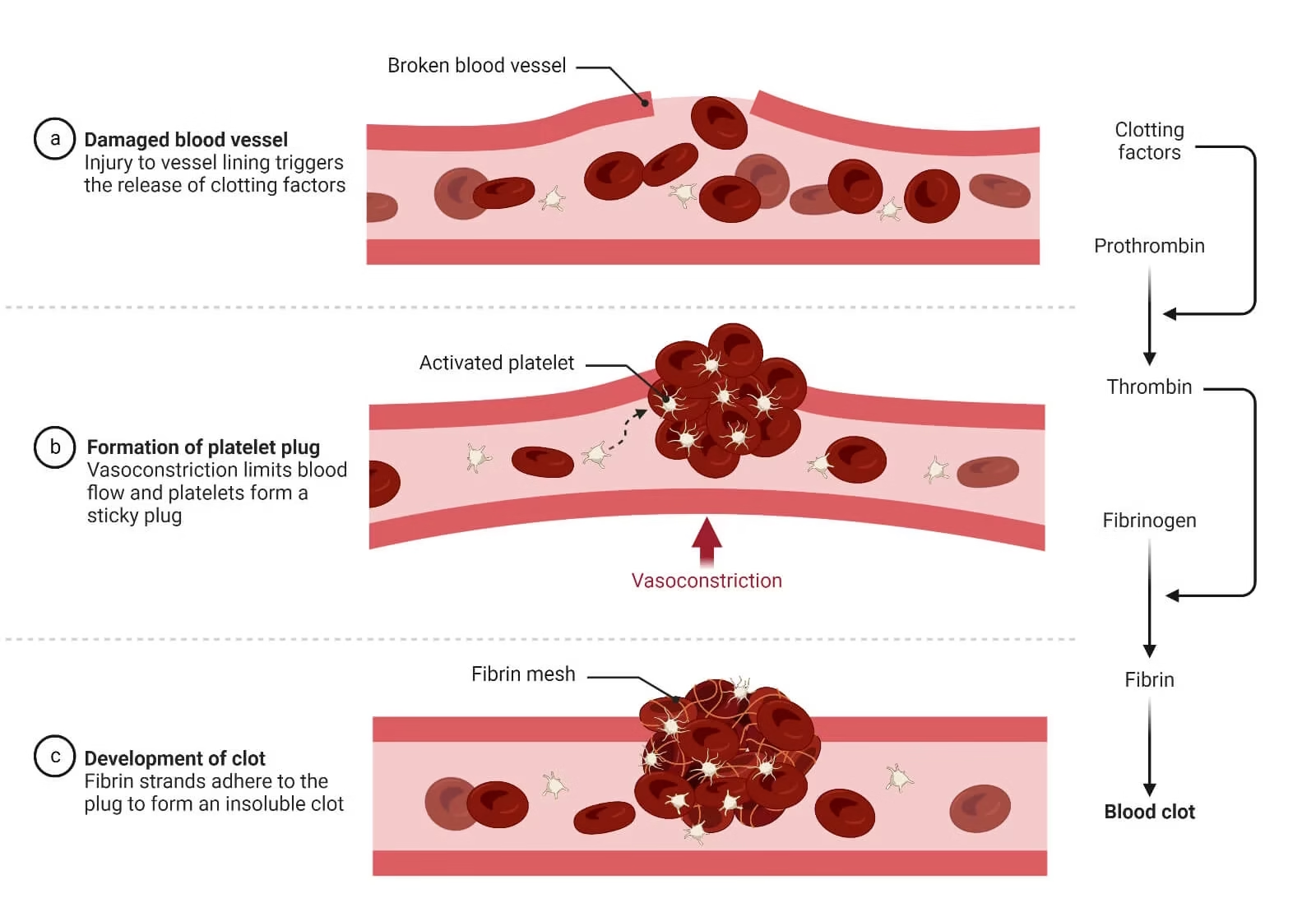
This step strengthens the platelet plug using fibrin threads.
What is the coagulation cascade?
It is a series of enzyme reactions where inactive clotting factors become active.
Coagulation Pathways
1. Intrinsic Pathway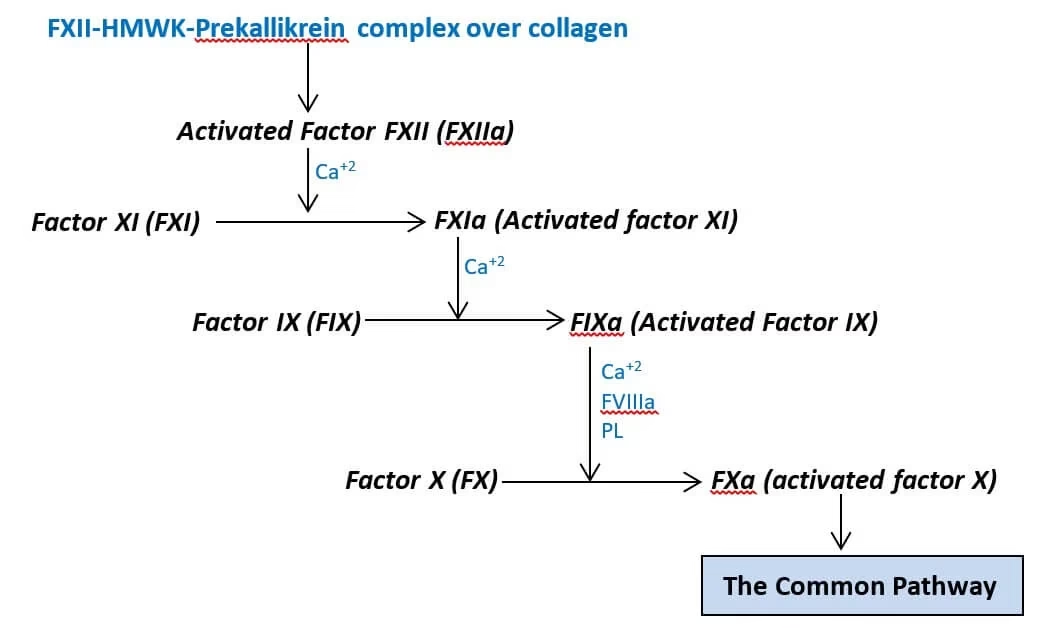
-
Starts inside the blood
-
Activated by contact with damaged surfaces
-
Important factors: XII, XI, IX, VIII
-
Tested by aPTT
2. Extrinsic Pathway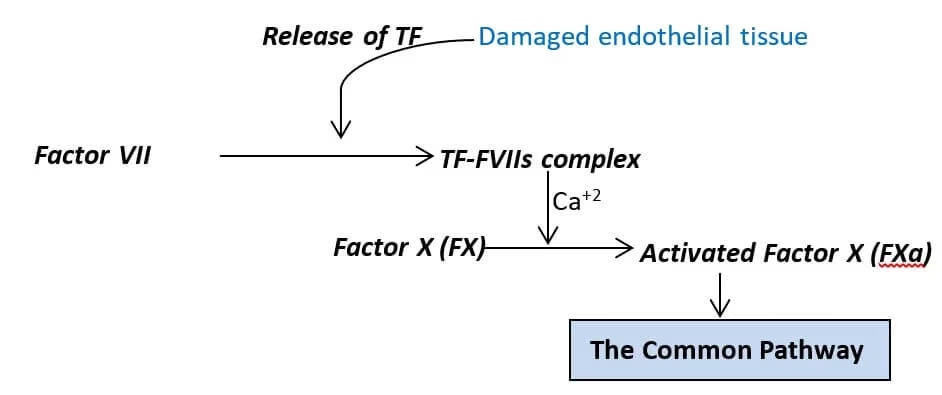
-
Starts from tissue injury
-
Involves tissue factor (Factor III)
-
Faster than intrinsic pathway
-
Tested by PT
3. Common Pathway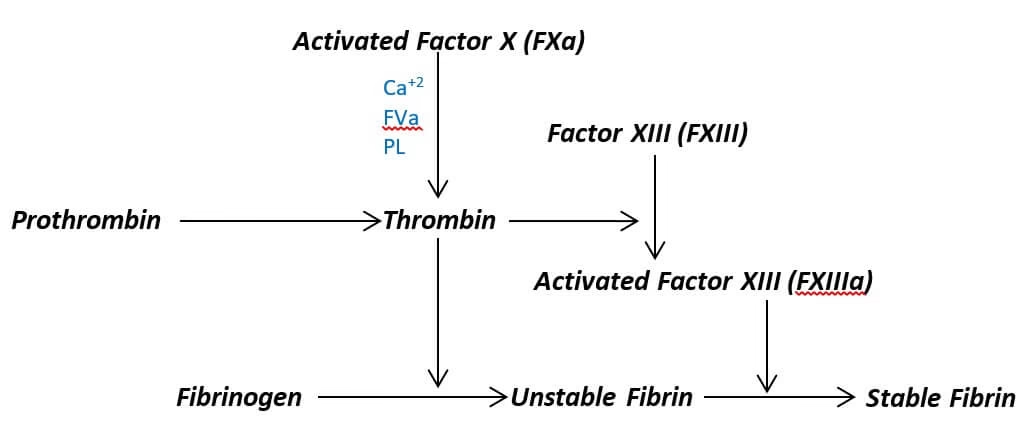
-
Both pathways meet at Factor X
-
Leads to:
-
Prothrombin → Thrombin
-
Fibrinogen → Fibrin
-
-
Fibrin forms a strong mesh around platelet plug
Vitamin K–dependent factors: II, VII, IX, X
Step 4: Clot Retraction and Vessel Repair
-
Platelets contract using actin and myosin
-
Clot becomes smaller and firmer
-
Pulls wound edges closer
-
Helps in healing of the blood vessel
Step 5: Fibrinolysis (Clot Removal)
Once healing is complete, the clot must be removed.
-
Plasminogen → Plasmin
-
Plasmin breaks fibrin into fibrin degradation products (FDPs)
-
Restores normal blood flow
This prevents unnecessary blockage of blood vessels.
Regulation of Hemostasis
1. Localized Activation of Clotting
-
Coagulation is activated only at the site of vascular injury.
-
Exposure of subendothelial collagen and tissue factor (Factor III) triggers platelet adhesion and coagulation cascade.
-
Intact endothelium away from injury inhibits clot formation.
2. Role of Vascular Endothelium
The endothelium plays a central regulatory role.
Antithrombotic Properties
-
Secretes prostacyclin (PGI₂) → inhibits platelet aggregation.
-
Releases nitric oxide (NO) → vasodilation and platelet inhibition.
-
Expresses thrombomodulin → binds thrombin and activates Protein C.
-
Heparan sulfate enhances antithrombin III activity.
Prothrombotic Role (at Injury Site)
-
Releases von Willebrand factor (vWF).
-
Expresses tissue factor initiating coagulation.
3. Natural Anticoagulant Systems
These prevent uncontrolled clot propagation.
a. Antithrombin III (AT III)
-
Inhibits thrombin (Factor IIa) and Factors IXa, Xa, XIa, XIIa.
-
Activity enhanced by heparin.
b. Protein C–Protein S System
-
Activated Protein C (APC) inactivates Factors Va and VIIIa.
-
Requires thrombomodulin and Protein S as a cofactor.
-
Vitamin K–dependent.
c. Tissue Factor Pathway Inhibitor (TFPI)
-
Inhibits Factor Xa–TF–VIIa complex.
-
Limits initiation of coagulation.
4. Regulation by Fibrinolytic System
-
Prevents persistence and spread of the clot.
Key Components
-
Plasminogen → Plasmin (via tPA and uPA).
-
Plasmin degrades fibrin into fibrin degradation products (FDPs).
Inhibitors
-
α2-antiplasmin inactivates plasmin.
-
PAI-1 inhibits tissue plasminogen activator (tPA).
5. Dilution and Clearance of Activated Factors
-
Continuous blood flow dilutes clotting factors.
-
Liver and reticuloendothelial system remove activated coagulation factors.
6. Feedback Control Mechanisms
-
Thrombin has dual roles:
-
Procoagulant: activates Factors V, VIII, XI.
-
Anticoagulant: activates Protein C via thrombomodulin.
-
-
Ensures balanced clot formation.
7. Platelet Regulation
-
Platelet activation is confined to damaged endothelium.
-
Intact endothelium prevents platelet adhesion.
-
Excess platelets are cleared by the spleen.
8. Clinical Correlation
-
Defective regulation → bleeding disorders (e.g., hemophilia).
-
Excessive regulation failure → thrombosis (DVT, stroke, MI).
-
Impaired fibrinolysis → persistent clots.
-
Excess fibrinolysis → bleeding.
Clinical Importance
1. Prevention of Excessive Blood Loss
-
Hemostasis rapidly forms a platelet plug and fibrin clot at the site of vascular injury.
-
Defective hemostasis results in prolonged bleeding, hemorrhage, and shock.
-
Clinically important in trauma, surgery, childbirth, and invasive procedures.
2. Maintenance of Normal Blood Flow
-
Under physiological conditions, anticoagulant and fibrinolytic systems keep blood in a fluid state.
-
Prevents unnecessary clot formation within intact blood vessels.
-
Failure leads to pathological thrombosis.
3. Role in Bleeding Disorders
-
Platelet disorders: Thrombocytopenia, platelet function defects → mucocutaneous bleeding.
-
Coagulation factor deficiencies:
-
Hemophilia A (Factor VIII deficiency)
-
Hemophilia B (Factor IX deficiency)
-
-
Vitamin K deficiency affects synthesis of clotting factors II, VII, IX, X.
4. Thrombotic Disorders
-
Excessive activation of coagulation leads to thrombosis.
-
Clinical conditions include:
-
Deep vein thrombosis (DVT)
-
Pulmonary embolism
-
Myocardial infarction
-
Ischemic stroke
-
-
Seen in atherosclerosis, immobilization, malignancy, and hypercoagulable states.
5. Importance in Liver Diseases
-
Liver synthesizes most coagulation factors.
-
Liver dysfunction causes bleeding tendencies due to reduced factor production.
-
Clinically assessed by PT/INR and APTT.
6. Role in Disseminated Intravascular Coagulation (DIC)
-
Widespread activation of coagulation → microthrombi formation.
-
Consumption of clotting factors and platelets leads to simultaneous bleeding and thrombosis.
-
Seen in sepsis, obstetric complications, trauma, malignancy.
7. Significance in Anticoagulant Therapy
-
Understanding hemostasis is crucial for the use of:
-
Heparin
-
Warfarin
-
Direct oral anticoagulants (DOACs)
-
-
Used to prevent and treat thromboembolic disorders while minimizing bleeding risk.
8. Importance in Diagnostic Medicine
-
Hemostatic tests help evaluate bleeding and clotting status:
-
Bleeding time
-
Platelet count
-
PT, INR
-
APTT
-
Fibrinogen levels
-
D-dimer
-
9. Role in Wound Healing and Inflammation
-
Fibrin clot acts as a temporary matrix for tissue repair.
-
Platelets release growth factors aiding wound healing.
MCQs
1. Hemostasis is best defined as:
A. Prevention of clot formation
B. Maintenance of blood pressure
C. Arrest of bleeding from damaged blood vessels
D. Dissolution of clots
2. Which factor initiates the extrinsic pathway of coagulation?
A. Factor XII
B. Factor VIII
C. Tissue factor
D. Factor IX
3. Which cell type plays a central role in regulating hemostasis?
A. Neutrophils
B. Platelets
C. Endothelial cells
D. Red blood cells
4. Prostacyclin (PGI₂) released by endothelium causes:
A. Platelet aggregation
B. Vasoconstriction
C. Inhibition of platelet aggregation
D. Activation of thrombin
5. Nitric oxide (NO) contributes to hemostatic regulation by:
A. Activating fibrinogen
B. Promoting platelet adhesion
C. Vasodilation and platelet inhibition
D. Activating factor X
6. Thrombomodulin is a receptor for:
A. Fibrinogen
B. Thrombin
C. Plasminogen
D. Factor Xa
7. Binding of thrombin to thrombomodulin leads to activation of:
A. Protein S
B. Protein C
C. Antithrombin III
D. Plasmin
8. Activated Protein C inactivates which clotting factors?
A. II and VII
B. IX and XI
C. Va and VIIIa
D. X and XII
9. Protein S acts as a:
A. Protease
B. Cofactor for Protein C
C. Platelet receptor
D. Fibrinolytic enzyme
10. Which anticoagulant is vitamin K–dependent?
A. Antithrombin III
B. Protein C
C. TFPI
D. Heparin
11. Antithrombin III primarily inhibits:
A. Platelets
B. Plasmin
C. Thrombin and factor Xa
D. Tissue factor
12. The anticoagulant effect of heparin is due to enhancement of:
A. Protein C
B. Protein S
C. Antithrombin III
D. TFPI
13. Tissue factor pathway inhibitor (TFPI) inhibits:
A. Factor XIIa
B. Factor Xa–TF–VIIa complex
C. Thrombin directly
D. Platelet aggregation
14. Which system limits clot size and duration?
A. Coagulation system
B. Platelet activation
C. Fibrinolytic system
D. Complement system
15. Plasminogen is converted to plasmin by:
A. Antithrombin
B. Protein C
C. Tissue plasminogen activator
D. Factor XIII
16. Major physiological activator of fibrinolysis is:
A. uPA
B. tPA
C. Thrombin
D. Factor X
17. Plasmin acts mainly on:
A. Platelets
B. Prothrombin
C. Fibrin
D. Tissue factor
18. Alpha-2 antiplasmin inhibits:
A. tPA
B. Thrombin
C. Plasmin
D. Factor Xa
19. PAI-1 inhibits:
A. Plasmin
B. Plasminogen
C. tPA
D. Fibrin
20. D-dimer is a marker of:
A. Platelet activation
B. Coagulation factor synthesis
C. Fibrin degradation
D. Endothelial damage
21. Normal blood flow prevents thrombosis mainly by:
A. Activating platelets
B. Diluting clotting factors
C. Increasing fibrin formation
D. Activating thrombin
22. Which organ clears activated clotting factors?
A. Kidney
B. Lung
C. Liver
D. Spleen
23. Intact endothelium prevents platelet adhesion by:
A. Releasing vWF
B. Expressing tissue factor
C. Releasing PGI₂ and NO
D. Activating factor XII
24. von Willebrand factor is released mainly from:
A. Platelets only
B. Endothelial cells
C. Liver cells
D. Plasma proteins
25. Excessive activation of coagulation with consumption of factors occurs in:
A. Hemophilia
B. Vitamin K deficiency
C. DIC
D. Thrombocytopenia
26. Disseminated intravascular coagulation results in:
A. Only bleeding
B. Only thrombosis
C. Both bleeding and thrombosis
D. No clinical effect
27. Which factor stabilizes the fibrin clot?
A. Factor VIII
B. Factor X
C. Factor XIII
D. Factor V
28. Thrombin has anticoagulant activity when it:
A. Activates fibrinogen
B. Activates platelets
C. Binds thrombomodulin
D. Activates factor XIII
29. Failure of natural anticoagulants leads to:
A. Hemorrhage
B. Thrombosis
C. Anemia
D. Leukopenia
30. Which is NOT a natural anticoagulant?
A. Antithrombin III
B. Protein C
C. Protein S
D. Fibrinogen
31. Primary hemostasis mainly involves:
A. Coagulation factors
B. Platelets and endothelium
C. Fibrinolysis
D. Liver function
32. Secondary hemostasis mainly involves:
A. Platelet plug formation
B. Coagulation cascade
C. Vasoconstriction
D. Endothelial repair
33. Which test assesses extrinsic pathway regulation?
A. APTT
B. Bleeding time
C. PT/INR
D. Platelet count
34. Which test reflects fibrinolysis?
A. PT
B. APTT
C. D-dimer
D. Bleeding time
35. Vitamin K deficiency affects synthesis of:
A. Platelets
B. Factors II, VII, IX, X
C. Factor XII only
D. Fibrinogen only
36. Protein C deficiency predisposes to:
A. Bleeding
B. Thrombosis
C. Anemia
D. Infection
37. Which molecule links coagulation and anticoagulation?
A. Plasmin
B. Thrombin
C. Factor XIII
D. Platelets
38. Platelet aggregation is inhibited by:
A. Thromboxane A₂
B. ADP
C. Prostacyclin
D. Collagen
39. Which condition shows excessive fibrinolysis?
A. DIC (late stage)
B. Hemophilia
C. Atherosclerosis
D. Polycythemia
40. TFPI is produced mainly by:
A. Platelets
B. Liver
C. Endothelium
D. Plasma cells
41. Which factor is most important in limiting clot spread?
A. Platelet count
B. Blood flow
C. Hemoglobin
D. RBC deformability
42. Natural anticoagulants act mainly to inhibit:
A. Platelet adhesion
B. Coagulation cascade
C. Vasoconstriction
D. Endothelial repair
43. Heparan sulfate enhances activity of:
A. Protein C
B. Antithrombin III
C. TFPI
D. Plasminogen
44. Endothelial damage shifts balance toward:
A. Anticoagulation
B. Fibrinolysis
C. Procoagulation
D. Vasodilation
45. Which is a marker of ongoing coagulation and fibrinolysis?
A. PT
B. INR
C. D-dimer
D. Platelet count
46. Platelet life span is approximately:
A. 1–2 days
B. 3–4 days
C. 7–10 days
D. 20–30 days
47. Removal of old platelets occurs mainly in:
A. Liver
B. Kidney
C. Spleen
D. Lung
48. Failure of fibrinolysis leads to:
A. Excess bleeding
B. Persistent clots
C. Anemia
D. Thrombocytopenia
49. Excess fibrinolysis results in:
A. Thrombosis
B. Hemorrhage
C. Hypertension
D. Polycythemia
50. Regulation of hemostasis ensures:
A. Continuous clotting
B. Continuous bleeding
C. Balance between clot formation and dissolution
D. Only platelet activation
Answer Key
-
C
-
C
-
C
-
C
-
C
-
B
-
B
-
C
-
B
-
B
-
C
-
C
-
B
-
C
-
C
-
B
-
C
-
C
-
C
-
C
-
B
-
C
-
C
-
B
-
C
-
C
-
C
-
C
-
B
-
D
-
B
-
B
-
C
-
C
-
B
-
B
-
B
-
C
-
A
-
C
-
B
-
B
-
B
-
C
-
C
-
C
-
C
-
B
-
B
-
C
