Introduction
- Determination of bacterial count is an important laboratory procedure used to estimate the number of microorganisms present in a sample.
- Bacterial counting helps in:
- Assessment of contamination
- Monitoring microbial growth
- Evaluation of sterilization and disinfection
- Clinical diagnosis
- Food and water microbiology
- Bacterial counting methods are broadly divided into:
- Total bacterial count
- Viable bacterial count
- Total count includes both living and dead bacteria, whereas viable count includes only living bacteria capable of growth and multiplication.
Total Bacterial Count
- Total bacterial count estimates the overall bacterial population within a sample, regardless of whether the cells are alive or dead.
- This measure is essential in contexts where bacterial biomass or general population dynamics are of interest, such as environmental studies, microbiome research, or quality control in industrial applications.
Methods for Total Bacterial Count
Direct Microscopic Count
- Direct Microscopic Count (DMC) is one of the simplest and oldest methods used for counting bacteria directly under the microscope.
- It is used to determine the total number of bacterial cells present in a given sample.
- This method counts all bacterial cells, including:
- Living bacteria
- Dead bacteria
- Because it counts both live and dead organisms, it is called a total bacterial count method.
- It is especially useful when rapid estimation of bacterial population is required without waiting for culture growth.
- DMC is commonly used in:
- Microbiology laboratories
- Milk bacteriology
- Water analysis
- Fermentation studies
- Industrial microbiology
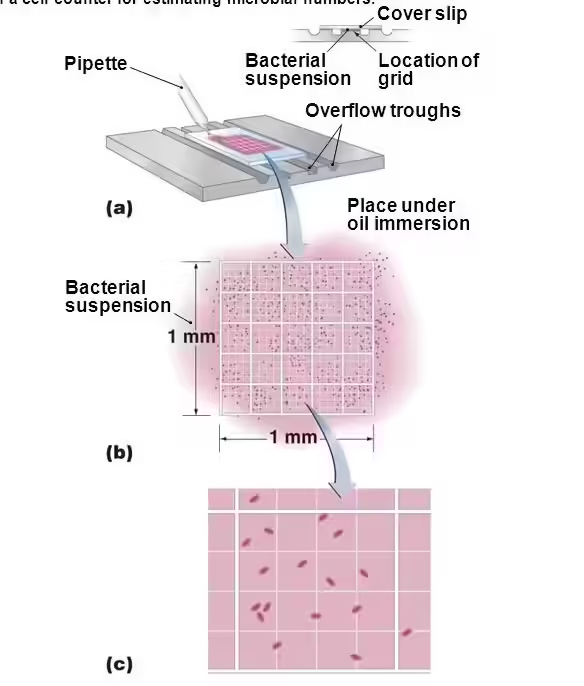
Principle
- A measured volume of bacterial suspension is placed in a special counting chamber.
- The chamber contains ruled squares of known dimensions.
- Bacteria present in selected microscopic squares are counted under the microscope.
- Since the area and depth of the chamber are known, the volume represented by each square can be calculated.
- From this, the number of bacteria per milliliter of sample is determined.
Counting Chamber Used in DMC
Common Counting Chamber
- Petroff-Hausser chamber
Structure of Counting Chamber
- It is a thick glass slide with:
- Central ruled grid
- Known square dimensions
- Fixed chamber depth
Chamber Depth
- Usually 0.02 mm
Grid Arrangement
- Large square divided into many small squares for counting.

Materials Required
- Bacterial suspension
- Petroff-Hausser counting chamber
- Coverslip
- Microscope
- Pipette
- Diluting fluid if required
Preparation of Sample
If Sample is Too Concentrated
- Dilution is required before counting.
Common Dilution
- 1:10
- 1:100
Importance of Proper Dilution
- Prevents overcrowding of bacteria
- Improves counting accuracy
Procedure
| Step | Procedure |
|---|---|
| 1 | Clean counting chamber and coverslip carefully |
| 2 | Place coverslip over chamber |
| 3 | Take well-mixed bacterial suspension |
| 4 | Introduce sample into chamber by capillary action |
| 5 | Allow bacteria to settle for a few minutes |
| 6 | Focus under microscope |
| 7 | Count bacteria in selected squares |
| 8 | Repeat counting in several fields |
| 9 | Calculate average count |
Microscopic Examination
Objective Used
- Usually oil immersion objective is used.
Counting Method
- Count bacteria in multiple squares.
- Average count is taken for calculation.
Counting Rule
- Count organisms touching:
- Upper boundary line
- Left boundary line
- Do not count organisms touching:
- Lower boundary line
- Right boundary line
Calculation of Bacterial Count
Formula
Number of bacteria per mL = Average number of bacteria per square×Dilution factor / Volume of one square
Observation
- All bacterial cells visible in the selected squares are counted.
- Both live and dead bacteria are included.
Advantages of Direct Microscopic Count
Major Advantages
- Rapid method
- No incubation required
- Immediate result obtained
- Useful for heavily contaminated samples
- Simple laboratory procedure
Flow Cytometry
- Flow cytometry is an advanced laboratory method used to analyze physical and chemical properties of cells or particles suspended in fluid.
- It allows rapid examination of thousands of cells individually within a few seconds.
- The technique is widely used in hematology, immunology, oncology, microbiology, and transplant medicine.
- In flow cytometry, cells pass one by one through a laser beam, and signals produced are recorded and analyzed by computer.
Principle
- Cells suspended in fluid pass through a narrow chamber in single file.
- Each cell passes through a laser beam.
- Laser light interacting with the cell produces:
- Forward scatter (FSC)
- Side scatter (SSC)
- Fluorescence signals
- These signals are detected and converted into electronic data.
Components
Fluidics System
- Transports cells in fluid stream.
- Ensures cells pass singly through laser beam.
Optical System
- Contains laser source, lenses, and filters.
- Illuminates cells and collects light signals.
Detection System
- Converts optical signals into electrical signals.
- Uses photodiodes and photomultiplier tubes.


Procedure
- Prepare cell suspension.
- Add fluorescent-labeled antibodies.
- Incubate sample.
- Wash excess reagent.
- Run sample through flow cytometer.
- Analyze signals by computer.
Data Presentation
Histogram
- Shows single parameter distribution.
Dot Plot
- Shows relationship between two parameters.


Applications
Hematology
- Leukemia and lymphoma diagnosis
Immunology
- CD marker analysis
Oncology
- Tumor cell analysis
Microbiology
- Cell counting
Transplantation
- Immune monitoring
Turbidity Measurement
- Turbidity measurement is a rapid method used for estimating the total number of bacteria present in a liquid culture.
- When bacteria multiply in broth medium, the medium becomes cloudy or turbid because bacterial cells remain suspended in the liquid.
- This turbidity is directly related to bacterial concentration.
- Since both living and dead bacterial cells contribute to cloudiness, this method gives total bacterial count rather than viable count.
Principle
- Suspended bacterial cells scatter light passing through the liquid medium.
- As bacterial concentration increases:
- Light transmission decreases
- Light scattering increases
- The degree of turbidity is proportional to bacterial population.
Instruments Used
Spectrophotometer
- Spectrophotometry is commonly used to measure turbidity.
- It measures optical density (OD) or absorbance.
Nephelometer
- Measures scattered light from bacterial suspension.

Procedure
| Step | Procedure |
|---|---|
| 1 | Prepare bacterial broth culture |
| 2 | Mix sample properly |
| 3 | Fill clean cuvette with bacterial suspension |
| 4 | Place cuvette in spectrophotometer |
| 5 | Select suitable wavelength |
| 6 | Record optical density |
Observation
- Clear broth indicates low bacterial count.
- Increased turbidity indicates increased bacterial count.
Interpretation
- Higher optical density indicates greater bacterial concentration.
- Optical density is compared with standard calibration curve to estimate bacterial count.
Calculation
- Optical density value is converted into bacterial concentration using standard reference curve.
Viable Bacterial Count
- Viable bacterial count is the method used to determine the number of living bacteria present in a sample.
- Only bacteria capable of growth and multiplication are counted.
- Dead bacteria are not included in this method.
- The result is expressed as CFU/mL (Colony Forming Units per milliliter).
- This method is important because each visible colony on culture medium is assumed to arise from one viable bacterial cell or a group of viable cells.
Methods for Viable Bacterial Count
Plate Count Method
Principle
- A bacterial sample usually contains very large numbers of bacteria, so direct plating may produce overcrowded colonies.
- Therefore, serial dilution of the sample is performed first.
- A measured quantity of diluted sample is inoculated on nutrient agar.
- During incubation, viable bacteria multiply and form visible colonies.
- Colonies are counted, and bacterial concentration is calculated.
Materials Required
- Sterile nutrient agar plates
- Sterile dilution tubes
- Sterile pipettes
- Inoculating spreader
- Incubator
- Bacterial sample
Serial Dilution Before Plate Count
Why Serial Dilution is Required
- To reduce bacterial concentration.
- To obtain countable colonies.
Common Dilutions
- 10⁻¹
- 10⁻²
- 10⁻³
- 10⁻⁴
Procedure of Plate Count Method
| Step | Procedure |
|---|---|
| 1 | Prepare serial dilutions of bacterial sample |
| 2 | Select suitable dilution |
| 3 | Transfer measured volume to sterile petri plate |
| 4 | Add sterile molten nutrient agar (for pour plate) or spread on agar surface (for spread plate) |
| 5 | Allow medium to solidify |
| 6 | Incubate at suitable temperature |
| 7 | Count colonies after incubation |
Types of Plate Count Method
1. Pour Plate Method
Procedure
- 1 mL diluted sample is placed in sterile petri dish.
- Molten agar at 45°C is added.
- Plate is gently rotated.
- After solidification, incubate.
Result
- Colonies develop:
- Inside agar
- On agar surface
2. Spread Plate Method
Procedure
- Small volume of diluted sample is placed on agar surface.
- Sterile spreader distributes sample evenly.
- Plate is incubated.
Result
- Colonies form only on surface.
Incubation
Temperature
- Usually 37°C for clinical bacteria
Duration
- 18–24 hours or according to organism
Colony Counting
Ideal Countable Range
- 30–300 colonies per plate
Why This Range is Used
- Less than 30 colonies → statistically unreliable
- More than 300 colonies → overcrowding and merging
Calculation of Viable Count
Formula
CFU/mL = Number of colonies × Dilution factor / Volume inoculated
Observation
- Each separate colony is counted.
- Mixed colony types may indicate contamination.
Interpretation
- More colonies indicate higher viable bacterial count.
- Colony morphology may also provide preliminary identification.
Advantages of Plate Count Method
Major Advantages
- Counts only living bacteria
- Highly accurate
- Widely accepted standard method
Most Probable Number (MPN) Method
Principle
- A sample is serially diluted and inoculated into multiple broth tubes.
- After incubation, tubes are examined for bacterial growth.
- The number of positive tubes in each dilution series is compared with standard MPN statistical tables.
- From this pattern, the most probable bacterial count is estimated.
Why It Is Called Most Probable Number
- The exact number of bacteria cannot be directly counted.
- Statistical probability is used to estimate the most likely number of viable organisms present.
Materials Required
- Sterile broth tubes
- Durham tubes (for gas detection if coliform testing)
- Pipettes
- Sample for testing
- Incubator

Procedure
| Step | Procedure |
|---|---|
| 1 | Prepare serial dilutions of sample |
| 2 | Inoculate measured volume into sets of broth tubes |
| 3 | Usually three sets of three tubes are used |
| 4 | Incubate at suitable temperature |
| 5 | Observe for growth, turbidity, acid, or gas production |
| 6 | Record number of positive tubes |
Arrangement of Tubes
Common Three-Tube Method
- First set: 10 mL sample
- Second set: 1 mL sample
- Third set: 0.1 mL sample
Observation
Positive Tube Shows
- Turbidity
- Gas formation
- Color change depending on medium
Negative Tube Shows
- Clear broth without reaction
Interpretation
- Positive tube pattern is written as three-digit combination.
Membrane Filtration
Principle
- A measured volume of liquid sample is passed through a membrane filter with very small pores.
- The membrane usually has pore size 0.45 µm, which retains bacteria.
- After filtration, the membrane is placed on nutrient agar or selective medium.
- Viable bacteria retained on the membrane grow into visible colonies during incubation.
Membrane Filter Used
Characteristics
- Thin sterile membrane
- Uniform pore size
Common Pore Size
- 0.45 micrometer
Function
- Retains bacteria while liquid passes through.

Apparatus Required
- Filtration unit
- Vacuum pump
- Membrane filter
- Sterile forceps
- Nutrient agar plate
Procedure
| Step | Procedure |
|---|---|
| 1 | Assemble sterile filtration apparatus |
| 2 | Place sterile membrane filter in holder |
| 3 | Pour measured liquid sample into funnel |
| 4 | Apply vacuum for filtration |
| 5 | Remove membrane using sterile forceps |
| 6 | Place membrane on culture medium |
| 7 | Incubate at suitable temperature |
| 8 | Count colonies formed |
Observation
- Colonies appear directly on membrane surface after incubation.
- Each colony represents one viable organism retained on filter.
Calculation
Formula
CFU per mL = Number of colonies counted / Volume of sample filtered
Advanced Methods and Viability Distinction Techniques
Practical Applications in Various Fields
Clinical Microbiology
- Used for bacterial count in:
- Urine samples
- Blood cultures
- Sputum samples
- Wound specimens
- Helps determine infection severity and monitor treatment response.
Water Analysis
- Used to assess microbial safety of drinking water.
- Common methods:
- Most Probable Number (MPN)
- Membrane filtration
- Detects coliform bacteria indicating fecal contamination.

Food Microbiology
- Used to detect bacterial contamination in:
- Milk
- Meat
- Vegetables
- Processed foods
- Ensures food safety and shelf-life monitoring.
Pharmaceutical Industry
- Used for sterility testing of:
- Injections
- Syrups
- Tablets
- Intravenous fluids
Dairy Industry
- Bacterial count determines milk quality and hygiene during processing.
Fermentation Industry
- Used to monitor bacterial growth during production of:
- Antibiotics
- Yogurt
- Organic acids
Research Laboratories
- Used for microbial growth studies and experimental analysis.
Public Health Microbiology
- Helps monitor outbreaks and environmental contamination.



